Ocular tuberculosis (OTB) is a rare form of tuberculosis. Diagnosis is usually presumptive based on epidemiology, clinical findings and positivity of immune response. The aim of the study was to characterize the OTB cases identified in Lisbon.
MethodsRetrospective study on OTB cases diagnosed in Lisbon from 2012 to 2015. The authors gathered data regarding demographic characteristics, diagnostic criteria, type of treatment as well as therapeutic results.
ResultsThere were 39 reported cases of presumed OTB which accounted for 12.2% of all reported TB cases. Patients had a mean age of 55.5 years. All had ocular manifestations (mainly uveitis). Diagnostic criteria included positive tuberculin skin test (TST) and/or Interferon Gamma Release Assay (IGRA). Culture testing or PCR could not be determined in any patient. Patients had OTB treatment for an average of 9.08 months. Clinical improvement was reported in 91.4% of treated patients. There were no relapses.
ConclusionThis is the first published series about OTB in Lisbon. All cases had a presumptive diagnosis made through TST and/or IGRA testing. Most of treated patients improved, with no signs of relapse and a median 29.9-month follow up. OTB seems to be a growing diagnosis and treatment may be beneficial even in patients with a presumptive diagnosis.
Tuberculosis (TB) continues to be one of the world's leading infectious causes of morbidity and mortality.1 It is estimated that in 2015 there were 10.4 million new TB cases worldwide of which 6.1 million were reported.1 In the same year Portugal had 1987 new TB cases, with an incidence of 19.2 cases per 100000 inhabitants, considered a low incidence rate.2 However in cities like Oporto and Lisbon the incidence was still higher than 20 cases/100000 (31/100000 in Oporto and 28.5/100000 in Lisbon).2
Ocular tuberculosis (OTB) incidence has varied widely over time. There were prospective studies reporting different OTB incidences, such as 1.46% in a Spanish study conducted in 1967,3 18% in another smaller Spanish study in 19974 and a 2.8% incidence of choroidal granulomas in a 2002 Malawian study.5
The prevalence of OTB as an aetiology of uveitis is variable over the world; it is most important in areas where tuberculosis is endemic where it can reach up to 10%. OTB can occur as a primary infection (in which the eye is the port of entry) or secondary to hematogenous spread.7–9 Disease is more common after reactivation of dormant lesions in the eye, frequently with unilateral clinical manifestations.9,10 On the other hand it can also occur as a hypersensitivity reaction to Mycobacterium tuberculosis antigens from a distant focus (like the lungs), causing inflammation in the eye despite the absence of the mycobacteria.8,9
Uveitis is the most common ocular manifestation of the disease and typically is a granulomatous uveitis. The most common presentation is posterior uveitis.8
The most typical lesions related to OTB are choroidal granulomas, occlusive retinal vasculitis and multifocal serpiginoid choroiditis.6,11
To obtain a definitive diagnosis a tissue sample or ocular fluid is needed to test acid-fast smears, mycobacterial cultures or PCR analysis.12–14 These samples are technically difficult to collect, impose risks on patients (including visual loss from cataract, retinal detachment, or infection12) and do not always provide a diagnosis due to low sensitivity.
According to the recently published Portuguese position paper on diagnosis and treatment management of ocular tuberculosis,15 when definitive diagnosis is not possible but there are clinical manifestations of OTB or any inflammation of the eye that does not respond to standard treatment (like corticosteroids), together with confirmed TB exposure (positive immune response established with a positive Tuberculin Skin Test (TST) and/or Interferon gamma release assay (IGRA) test depending on the immune status of the patient) or evidence of healed or active tubercular lesion on chest X-ray or CT scan of the lungs, OTB can be presumed and treatment should be offered.12,15,16
Gupta and colleagues proposed a classification of intraocular tuberculosis (IOTB) cases into confirmed, probable and possible OTB (Table 1).11
IOTB diagnosis proposed classification.11
| OTB diagnosis | |
|---|---|
| Confirmed IOTB (both 1 and 2) | 1. At least one clinical sign suggestive of IOTB |
| 2. Microbiological confirmation of Mycobacterium tuberculosis (MTB) from ocular fluids/tissues | |
| Probable IOTB (1, 2, and 3 together) | 1. At least one clinical sign suggestive of IOTB (and other etiologies excluded) |
| 2. Evidence of chest X-ray consistent with TB infection or clinical evidence of extraocular TB or microbiological confirmation from sputum or extraocular sites | |
| 3. At least one of the following: a. Documented exposure to TB b. Immunological evidence TB infection | |
| Possible IOTB (1, 2, and 3 together) (or 1 and 4) | 1. At least one clinical sign suggestive of IOTB (and other etiologies excluded) |
| 2. Chest X-ray not consistent with TB infection and no clinical evidence of extraocular TB | |
| 3. At least one of the following: a. Documented exposure to TB b. Immunological evidence TB infection | |
| 4. Evidence of chest X-ray consistent with TB infection or clinical evidence of extraocular TB but none of the characteristics given in 3 | |
The drug regimens used for extrapulmonary TB are similar to those of pulmonary TB, with a four drug anti-tuberculosis treatment (ATT) with isoniazid, rifampicin, pyrazinamide and ethambutol for 8 weeks followed by isoniazid with rifampicin for four to 10 months.7,10
The use of ethambutol, as is well known, can cause optic neuropathy. This is a first choice drug to treat OTB but it should be used with caution and with ophthalmologic observation at least every two months.15
Manousaridis et al.12 believed that a six-month therapy was enough to treat OTB without risking ocular damage as a side effect of the treatment. Agrawal et al.17 found that a nine month or longer treatment had a higher success rate.
The 2017 Portuguese position paper15 recommends 6 to 9 months of treatment.
ObjectiveThe aim of this study was to characterize the recent OTB cases identified in Lisbon.
MethodsStudy designThis was a retrospective study. Medical records on all individuals diagnosed with OTB in a four-year period (from 2012 to 2015) and under follow-up in Centro de Diagnóstico Pneumológico Dr. Ribeiro Sanches (CDP) were reviewed. All data were processed anonymously according to the institution's privacy policy.
SettingAll TB patients in Portugal are referred to specialized centres according to residence address. This allows for correct epidemiology control.
Since 2012, CDP has been the reference centre for TB in the Lisbon, Loures and Oeiras counties of the district of Lisbon with a population of 1380000 in its influence area.
Ophthalmologists refer all cases with suspected OTB after exclusion of the most common causes of ocular inflammation and with no response or recurrence to standard treatment (usually including steroids) to CDP.
In CDP, patients were tested for immune response by TST and/or IGRA test depending on their immune status and evaluated with a symptom questionnaire to exclude pulmonary TB. When there was immunological evidence of TB infection and/or there was evidence of healed or active pulmonary tubercular lesion on chest X-ray or CT scan, OTB treatment was offered. At 2 months of treatment all patients were evaluated for the possibility of changing treatment to continuation phase. Although it was not always possible (due to scheduling difficulties), most of our patients had an evaluation by the referring ophthalmologist at that period.
Ophthalmologic re-evaluation generally took place after the first 6 months of treatment and every 3 months thereafter and depending on their clinical observation as well as on patient's symptoms it was decided if the treatment needed to continue or if it could be stopped.
Data collection and analysisInformation regarding demographic characteristics, other comorbidities and history of prior pulmonary TB was collected. Diagnostic criteria was characterized according to ocular manifestations, response to anti-inflammatory treatment, TST or IGRA testing, other diagnostic tests if available and extension of disease. Treatment was recorded according to medication regimen and duration and therapeutic results.
ResultsThere were 39 suspected cases of OTB sent to our Centre (12.2% of all reported TB cases). Thirty two of them were considered “possible OTB” and 7 were considered “probable OTB” due to radiological changes consistent with healed pulmonary tuberculosis (3 of them with visible Gohn's complex, one with residual thickening pleura and the other 3 with fibrocicatricial sequelae of the lung apex). All of them were offered treatment. Patients were predominantly females (22 females vs 17 males), with a mean age of 55.5 years and mostly of Portuguese origin (35 of 39 patients).
Two patients had auto-immune diseases (primary biliary cirrhosis (1) and rheumatoid arthritis (1)). HIV infection was tested in all patients but only one patient had co-infection HIV-B Hepatitis. Four patients were immunosuppressed for other reasons (diabetes (3) and cancer (1)).
Ten patients reported history of TB, including pulmonary (4), miliary (1), osseous TB (1) and childhood primary infection (4). Only two reported a recent (<2 years) contact with other TB patients.
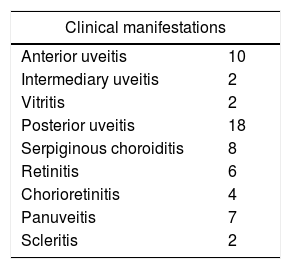
All of our patients were referred by Ophthalmologists for chronic inflammatory relapsing ocular diseases not responding to treatment after exclusion of more common etiologies. Table 2 cites ocular clinical manifestations reported.
The majority had OTB symptoms in both eyes (74% (29)) without prevalence of one eye over another in unilateral disease.
Diagnostic criteria included positive TST and/or IGRA. Thirty two (82%) patients had a TST test. The TST mean result was 22.56mm. Four TST were negative, all of them had positive IGRA.
IGRA was tested in 37 patients and was positive in all of them. Subjects not tested with IGRA were two immunosuppressed patients (DM type II and corticotherapy > 15mg of prednisolone or equivalent) with TST results >20mm. Culture testing or PCR were not determined in any patient.
Pulmonary tuberculosis was investigated by symptom questionnaire in all patients, coupled with chest X-ray (in 15 subjects) or CT scan (24 subjects) and when in doubt with sputum smear (seven subjects). All the 39 cases of OTB did not have other infectious locations identified (intra or extrapulmonary).
Table 3 presents anti-tuberculosis treatment of all patients. All of our patients had a two month initiation phase treatment, mostly with Isoniazid (H), Rifampicin (R), Pyrazinamide (Z) and Ethambutol (E), however in some patients Ethambutol was suppressed or substituted with Levofloxacin due to concerns with optic neuropathy. The continuation phase was comprised of only two drugs (Isoniazid (H) and Rifampicin (R) or Rifabutin (Rb)) (Table 3). The choice of initiation phase drugs did not affect the overall duration of treatment. There were no significant side effects reported.
Patients had OTB treatment for an average of 9.08 months (minimum of six months and maximum of 16 months). Considering length of treatment, 64.7% of the patients had treatment for at least nine to 12 months. The patient that had a 16-month course of treatment was a HIV positive patient with a bilateral pan-uveitis and the duration was discussed with the ophthalmologist.
Thirty five patients have completed treatment with an average follow-up period of 29.9 months. Three patients dropped out (two for lack of adherence to treatment and one for intolerance to the medication). One patient died while on treatment (after admission to an Intensive Care Unit for a non-related acute episode).
Clinical improvement (based on an ophthalmologist observation and patient's symptoms) was reported in 91.4% of the patients that completed treatment. Only three of the patients treated had no improvement. There were no relapses.
Discussion and conclusionThere is growing awareness of OTB with improved referral by ophthalmologists.
The diagnosis is usually presumptive because culture testing or PCR are usually very difficult to execute due to difficulty in obtaining specimens. Typical ocular manifestations like uveitis should raise suspicion for ophthalmologists. After exclusion of other inflammatory diseases, OTB should be suspected. A positive TST/IGRA must be sought.
This is one of the largest published case series of OTB in a low endemic country12,17–21 and the first published series about OTB in Lisbon, Portugal, however it is not without some limitations.
It is not a nation-wide study and we report cases from the Lisbon district alone. However, this is a very large district in one of the few metropolitan areas of Portugal, with an influence area of population of over one million. It is also our purpose to increase awareness of what is probably an underdiagnosed disease, especially in intermediate to low-incidence areas like other smaller centres in this and other countries. This was, however, a retrospective study on OTB diagnosed from 2012 to 2015. Another interesting study would be to design a prospective study for diagnosis of OTB in other TB patients.
Other limitations include the fact that OTB diagnosed cases were not confirmed by ocular samples due to technical difficulties and risk to the patients and so we report probable and possible OTB cases according to Gupta et al.11 This reflects the real-world situation; most cases are not confirmed due to technical difficulty. As well as this, decision when to stop the therapy was based on a subjective opinion (symptoms and clinical observation by ophthalmologist).
All of our patients had ocular manifestations suggesting OTB and TST and IGRA testing were essential for presumed diagnosis.
According to our data OTB should be suspected as a possible cause of uveitis after exclusion of the most probable causes, even if the patient does not have a history of tuberculosis or risk factors like autoimmune diseases or HIV infection.
All of our patients had only ocular symptoms with uveitis as the main manifestation.
As opposed to what has been reported in literature, most of our patients had bilateral ocular manifestations. This can possibly be explained by a secondary infection of the eyes through hematogenous spread.
Pharmacological therapy was mainly with standard regimen (HRZE). The use of Ethambutol was safe, with 85% of the patients including this drug in their treatment and there were no reports of optic neuropathy. The length of treatment was always discussed with ophthalmologists and was based on clinical improvement. In our series the length of treatment was greater than advocated in the most recent literature. One of the reasons could have been that this is a rare condition and the clinicians preferred to have a particularly zealous approach to avoid relapses. Also, at that time there were no guidelines available; the Portuguese recommendations referring to length of treatment16 were only published in 2017. In our experience nine months seems to have the best results without major side effects.
The majority of patients treated had good results with no signs of relapse with a median 29.9 months follow up.
In conclusion OTB seems to be a growing diagnosis and treatment improves quality of life of the majority of patients even with a presumptive diagnosis.
FundingThis research did not receive any specific grant from funding agencies in the public, commercial, or not-for-profit sectors.
Conflicts of interestThe authors have no conflicts of interest to declare.
All authors had contributed to the manuscript.