Since Hypersensitivity Pneumonitis (HP) categorization in fibrotic and nonfibrotic/inflammatory types seems to be more consistent with the distinctive clinical course and outcomes, recent international guidelines recommended the use of this classification. Moreover, fibrotic subtype may share immunogenetic and pathophysiological mechanisms with other fibrotic lung diseases.
AimTo investigate HLA -A, -B, -DRB1 and TNF-α –308 gene polymorphisms among fibrotic and nonfibrotic HP patients due to avian exposure, also in comparison with asymptomatic exposed controls.
MethodsWe prospectively enrolled 40 HP patients, classified as fibrotic or nonfibrotic/inflammatory, and 70 exposed controls. HLA and TNF-α polymorphisms were determined by polymerase chain reaction–sequence specific primer amplification.
ResultsWhile HLA alleles were not associated to HP susceptibility, fibrotic HP patients showed increased frequencies of HLA A*02 (46.7% vs 25.7%; OR=2.53, p = 0.02) and HLA DRB1*14 (10.0% vs 0.7%; OR=15.44, p=0.02) alleles when compared with exposed controls, although not statistically significant after correction for multiple comparisons. TNF-α G/G genotype (associated with low TNF-α production) frequencies were significantly increased among the non-fibrotic/inflammatory HP patients comparatively to fibrotic presentations (88% vs 60%; RR=0.44; p=0.04) and controls (88% vs 63%, OR 4.33, p=0.037). Also, these patients had a significantly increased frequency of the G allele (94.0% vs 73.3%, RR=0.44, p=0.01), while fibrotic HP patients predominantly presented the A allele (26.7% vs 6.0%, RR=2.28, p=0.01).
ConclusionsOur results support the hypothesis that fibrotic and non-fibrotic HP subtypes exhibit a distinct profile of TNF-α and HLA polymorphisms, which may be relevant to predict disease course and better define treatment strategies.
Hypersensitivity pneumonitis (HP) is an inflammatory and/or fibrotic disease affecting the lung parenchyma and small airways, resulting from an immune-mediated reaction induced by inhalation of organic or chemical antigens in susceptible individuals.1–5 Although frequently unidentified,6,7 several antigens are known to cause the disease, avian proteins being one of the most commonly involved.2,3 The immune response after antigen exposure among sensitized individuals include antigen-specific IgG antibodies and a CD4+ T-helper cell type 1 (Th1) and CD8+ T-cytotoxic immune response,8,9 associated with the characteristic lymphocytic and granulomatous inflammation.3,10,11 Later, and especially during the fibrotic phase, neutrophilic inflammation seems to play a role.12,13 Additionally, a T cell regulatory cells dysfunction and T-helper cell polarization underlines an exaggerated immune response and fibrotic response.14–17
Human leukocyte antigen (HLA) genes are located in the short arm of chromosome 6 and are essential in antigen presentation and immunoregulation.18 They encode two distinct classes of cell surface molecules: major histocompatibility complex (MHC) I and II,19 which provide the key surface molecules upon which specific CD8+ and CD4+ T lymphocytes, respectively recognize foreign antigens. Several studies support that HLA gene polymorphisms, especially those belonging to class II, are implicated in HP susceptibility, namely HLA-DR3 in Caucasians and HLA-DR7 and certain HLA-DRB1/DQB1 haplotypes in Hispanic patients with pigeon breeder's disease.20–24
On the other hand, cytokines are vital for cellular communication and key elements in lung immune response modulation.25 Certain polymorphisms in the promoter regions of cytokines genes have functional significance and may be related to HP clinical expression.25,26 Tumor necrosis factor alpha (TNF-α) is a proinflammatory cytokine encoded by MHC class III region that has been found to be involved in experimental models of HP and increased among HP patients.27–30 Moreover, certain TNF-α gene polymorphisms have been also associated to HP.23,24,31,32
According to recent guidelines,5 HP classification in fibrotic and nonfibrotic/inflammatory types is more consistently associated with the distinctive clinical course and outcomes.33,34 In fact, fibrosing interstitial lung diseases may share common immunogenetic pathways.35 Since polymorphisms at HLA class I and II loci, and position -308 of the TNF-α gene promoter region have been suggested to be involved in the fibrotic lung diseases pathogenesis,36–40 we investigated HLA -A, -B and -DRB1 and TNF-α –308 gene polymorphisms among fibrotic and nonfibrotic HP patients due to avian exposure, also in comparison with asymptomatic exposed controls.
Material and methodsStudy design and patient selectionAn observational prospective single-center case-control pilot study enrolled forty consecutive HP patients due to avian exposure and seventy asymptomatic individuals exposed to avian antigens, genotyped for HLA and cytokine polymorphisms.26 Since this is a convenience sample, no a-priori sample size calculations were performed. All participants were native Portuguese, Caucasians, and unrelated. Written informed consent was obtained from the participants and the study was approved by the Ethics Committee of our institution.
A multidisciplinary team (MDT) including pulmonologists, radiologists and pathologists 41,42 was involved in the diagnosis that accomplished the following criteria:43 1) avian exposure determined by direct questionnaire, 2) consistent HP clinical and radiological features, 3) bronchoalveolar lavage (BAL) lymphocytes > 40%, 4) in those patients whose diagnosis was not established by clinical, radiological and BAL features, surgical lung biopsy was performed (n=11). Patients were further classified as fibrotic, i.e. patients with radiological and/or histopathological evidence of fibrosis, or non-fibrotic/inflammatory cases, i.e. patients without radiological and/or histopathological evidence of fibrosis.5
DNA extractionGenomic DNA was isolated from anticoagulated venous blood samples by phenol precipitation followed by digestion with proteinase K a using Puregene DNA Isolation Kit (Gentra Systems, Minneapolis MN).
HLA genotypingAll individuals were genotyped for alleles at the HLA -A, -B and -DRB1 loci. The HLA genotyping was performed by Polymerase Chain Reaction – Sequence Specific Primers, (PCR-SSP), (Olerup SSP, GenoVision VertriebsgesmbH, Vienna, Austria). In this method, the pairs of the chosen primers are sequence specific, only amplifying when the terminal 3’ of the primer is complementary to the target sequence and alleles are determined by the presence of the specific PCR product by agarose gel electrophoresis stained by ethidium bromide.
Tumour necrosis factor (TNF-)-α genotypingPolymorphisms located in –308 bp promoter/enhancer region of TNF-α44,45 were determined by PCR-SSP amplification using the Cytokine Genotyping Tray (One Lambda, Inc., Canoga Park, CA). This analysis included positive and negative controls. All PCR products were fractionated electrophoretically in 2% agarose-gel and visualized by ethidium bromide staining and UV light.
Statistical analysisHLA allele frequencies and TNF-α genotypes were determined by direct counting for patients and controls. The χ2 test or Fisher exact test, when appropriate, were used to assess association of various alleles with disease susceptibility (HP patients vs controls) and fibrotic/non-fibrotic forms (i.e, HP fibrotic vs. HP non fibrotic, HP fibrotic vs. controls, HP non fibrotic vs. controls, HP (non fibrotic + fibrotic) vs. controls). To quantify these possible associations, Relative Risk (RR) – for comparisons between patients´ subgroups (fibrotic and non fibrotic) –, Odds Ratio (OR) – when compared HP patients (both fibrotic and non fibrotic) with exposed controls – and respective confidence intervals (C.I) at 95% were calculated. We compare each allele (or genotype) frequency against remaining alleles (or genotypes) frequencies. Hence, p values were corrected for the multiple comparisons (pc) using the formula pc= 1 – (1 – p)n,46 with n the number of alleles at each locus or the number of genotypes for the cytokine (n=18 for A*; n=23 for B*; n=12 for DRB1*; n = 3 for TNF-α). Lung functional and BALF parameters in patients’ genotypes subgroups were compared through Kruskal-Wallis test. Hardy-Weinberg equilibrium in the control group for HLA frequencies were tested with ‘hwe.hardy()’ function from ‘gap’ package.47 For all statistical tests performed, two-sided p-values lower than 0.05 were considered statistically significant, mainly for pc. All analysis was performed within R Studio (version 1.3.959),48 a computing environment for R (version 3.6.1) 49 programming language.
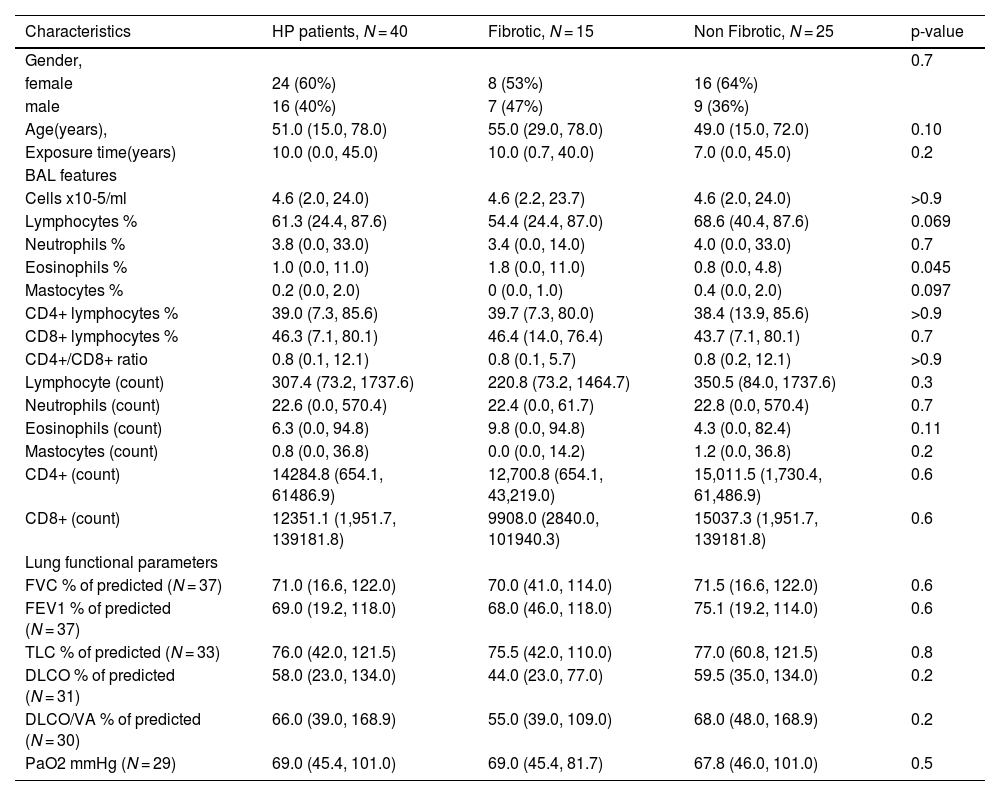
ResultsBaseline featuresAmong the forty enrolled HP patients, 15 were classified as fibrotic and 25 as non-fibrotic/inflammatory. The demographic and clinical characteristics of the HP cases are described in Table 1. No statistically significant differences were found concerning demographic and clinical features between the two patient's groups, except for BAL eosinophils which were higher in fibrotic than in non-fibrotic patients (1.8 % vs 0.8%; p=0.045). Healthy controls and HP patients presented no significant differences concerning gender distribution (females 65% vs 60%, p=0.69) and median exposure time (6 vs 10 years, p=0.10), but they differ on median age being the controls younger than the patients (34.3 vs 51 years-old; p<0.001).
Clinical and demographic characteristics of HP patients.
Statistics presented: median (minimum, maximum); n (%).
Statistical tests performed: Wilcoxon rank-sum test; chi-square test of independence; Fisher's exact test.
HLA allele frequencies did not show statistically significant deviations from Hardy-Weinberg equilibrium in the control group (data not shown). In this series, no particular HLA allele frequency differ between HP patients and exposed controls (Supplementary Tables 1-3). However, when comparing fibrotic HP patients with exposed controls (Supplementary Tables 4-6), HLA A*02 (46.7% vs 25.7%; OR=2.53, p=0.02) and HLA DRB1*14 (10.0% vs 0.7%; OR=15.44, p=0.02) alleles were more frequent among this HP subgroup. No statistically significant differences were found in HLA A, B, DRB1 allele frequencies between non-fibrotic/inflammatory HP patients and controls (Supplementary Tables 7-9). After correction for multiple comparisons, no statistically significant results were found (pc>0.05).
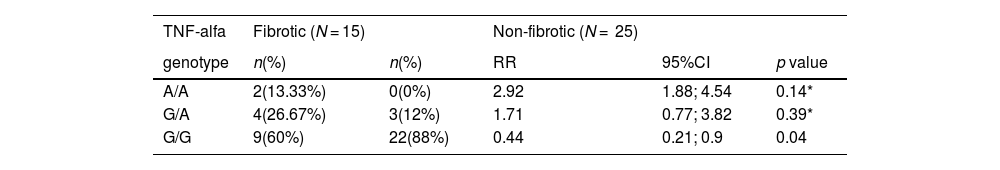
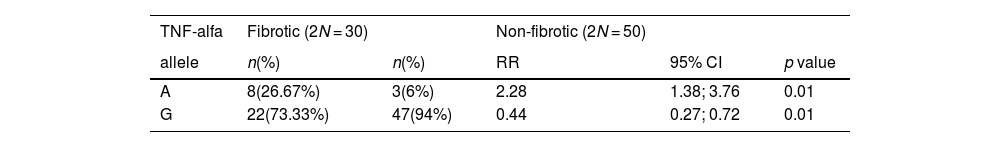
Tumour necrosis factor (TNF-)-α analysisAllele distributions of TNF-α (-308 A/G) revealed no statistically significant deviations from Hardy-Weinberg equilibrium in the control group (data not shown). TNF-α G/G genotype (associated with low TNF-α production) frequencies were significantly increased among the non-fibrotic/inflammatory HP patients comparatively to fibrotic presentations (88% vs 60%; RR=0.44; p=0.04) (Table 2) and controls (88% vs 63%, OR 4.33, p=0.037). When comparing allelic frequencies between non-fibrotic/inflammatory and fibrotic HP subgroups (Table 3), the first showed a significant increase in the frequency of the TNF-α G allele (94.0% vs 73.3%, RR=0.44, p=0.01), while the later presented the TNF-α A allele more frequently (26.7% vs 6.0%, RR=2.28, p=0.01). No differences were found considering TNF-α genotypes between fibrotic HP and controls (data not shown).
Genotypes for TNF-alfa cytokine expression in fibrotic versus non-fibrotic HP patients.
| TNF-alfa | Fibrotic (N = 15) | Non-fibrotic (N = 25) | |||
|---|---|---|---|---|---|
| genotype | n(%) | n(%) | RR | 95%CI | p value |
| A/A | 2(13.33%) | 0(0%) | 2.92 | 1.88; 4.54 | 0.14* |
| G/A | 4(26.67%) | 3(12%) | 1.71 | 0.77; 3.82 | 0.39* |
| G/G | 9(60%) | 22(88%) | 0.44 | 0.21; 0.9 | 0.04 |
n(%) number of patients (percentage).
RR relative risk.
95%CI 95% confidence interval.
Frequencies for TNF-alfa alleles in fibrotic versus non-fibrotic HP patients.
| TNF-alfa | Fibrotic (2N = 30) | Non-fibrotic (2N = 50) | |||
|---|---|---|---|---|---|
| allele | n(%) | n(%) | RR | 95% CI | p value |
| A | 8(26.67%) | 3(6%) | 2.28 | 1.38; 3.76 | 0.01 |
| G | 22(73.33%) | 47(94%) | 0.44 | 0.27; 0.72 | 0.01 |
n(%) number of alleles (percentage).
RR relative risk.
95%CI 95% confidence interval.
In this study, where we evaluated MHC class I/II and the TNF-α –308 gene polymorphisms in 40 patients with HP due to avian exposure, we found that those classified as nonfibrotic/inflammatory, according to the ATS/JRS/ALAT guidelines, had a significantly increased frequency of the TNF-α G/G genotype (associated with low TNF-α production), comparatively to fibrotic cases and asymptomatic exposed controls. Furthermore, while G allele was more commonly found among nonfibrotic HP patients, the A allele showed to be predominant in those classified as fibrotic.
Heterogeneous clinical presentation and outcomes are typical features of HP, that varies from spontaneous resolution to progressive lung function decline and respiratory failure. Whereas nonfibrotic or inflammatory subtype may have a benign course, fibrotic HP often present a progressive fibrotic phenotype compared to other fibrotic lung diseases.35 Definitely, similarities with IPF have been reported to be poor prognosis factors particularly the presence of usual interstitial pneumonia (UIP)-like pattern and absence of BAL lymphocytosis.50–54
Polymorphisms of the promoter regions of certain pro-inflammatory cytokines, such as IL-6 and IFN- γ may underlie the clinical heterogeneity of this disease.25,26 The results of the current study further extend these findings since a TNF-α polymorphism, consisting of guanine (G) to adenine (A) substitution at position –308 in the promoter region, previously associated with increased risk of developing pulmonary fibrosis in English, Italian and Australian populations,55,56 tended to be increased in the HP patients’ subgroup classified as fibrotic.
Experimental and pathological evidence supports the role of TNF-α in HP pathogenesis. In experimental mice models, HP provoked by the actinomycete Faenia rectivirgula was accompanied by an increase in TNF-α levels, while the use of an antibody against TNF-α was associated with HP resolution.30 Furthermore, TNF-α seems to promote lung injury,57 leukocyte margination and associates to fibroblast proliferation.58 In animal models, this proinflammatory cytokine was found to be overexpressed and considered essential for the development of lung fibrosis.59 In human studies, TNF-α also showed to be overexpressed in the fibrotic lung, increasing fibroblast response.60,61 High levels of this cytokine have been found in patients with HP.27–29 Since the allele A of the -308 TNF-α gene polymorphism is associated with increased mRNA transcription and higher levels of this cytokine than the G allele,32,62 the association found in our study corroborates the reported findings. Although our results require further validation, this polymorphism may possibly be a clinically useful biomarker to stratify HP patients and to select the best treatment strategy, i.e. immunosuppressants or antifibrotics, according to disease features.35
Regarding HLA polymorphisms, and despite there is some evidence indicating an association between HLA polymorphisms such as HLA-DR3, HLA-DR7, HLA-DQ3 and HLA-DRB1*04 and HP susceptibility,20–24 we did not find any statistically significant results in our series. Comparing our findings to previous published literature is hindered by the variability of the genetic backgrounds, different disease subtypes and distinctive exposures addressed in different studied populations. However, our results showed that A*02 and DRB1*14 HLA polymorphisms tended to be associated to an increased risk of developing a fibrotic HP. Evidence on the possible role of HLA in fibrotic lung diseases susceptibility is contradictory.23,36,38,39 Falfán-Valencia et al 37 reported that the HLA DRB1*1402 allele was significantly increased among IPF patients. Although the mechanisms underlying this finding are still to be elucidated, in an experimental model of interstitial fibrosis class II MHC expression in type II alveolar epithelium may occur in the early stage of cellular injury, followed by inflammatory infiltration;63 Moreover, HLA class II molecules are also expressed in alveolar epithelial cells from IPF lungs.64 Concerning HLA A*02, no published literature was found supporting the association with fibrosing lung diseases. However, in a Chinese population, and despite no difference in HLA allele frequencies, the frequency of HLA-A*02-DRB1*04 haplotype was significantly higher in IPF patients.40 Due to the small sample size, our work did not allow haplotype frequencies estimation.
The main limitation of the present study is the small sample size that hinders a more robust statistical analysis. Additionally, other factors that potentially influence disease subtype were not taken into consideration. Examples include other genetic features such as chemokines or antigen exposure patterns.65,66 Only patients with avian related HP were addressed in this study, which limits the generalization of the results to other HP types. However, since the relevance of a specific antigen in the disease course is still to be clarified,7,67 a more homogeneous sample eliminated the bias caused by the influence of antigen type. Other strengths of this work include the prospective follow-up of the participants and the fact of the HP patients were diagnosed by the same multidisciplinary team and followed-up by the same experienced pulmonologists from our reference centre.
Overall, these results besides contributing to a better understanding of fibrotic HP, support the hypothesis that progressive fibrosing interstitial lung diseases share immunogenetic and pathophysiological mechanisms, which may have clinical and therapeutic implications for HP patients, since treatment strategies may influence the natural course of the disease, often unknown at diagnosis.4,35
We acknowledge M. Beltrão, MSc for the contribution on the bronchoalveolar lavage analysis.