To investigate the perspective of physicians treating chronic airway diseases on the importance of device and substance characteristics influencing the compliance of patients with chronic obstructive airways diseases.
ObjectiveWe surveyed physicians... perspective on the impact of device and substance characteristics on patients... compliance.
MethodsThis study was carried out by running a structured questionnaire, to a total of 144 physicians, conducting personal interviews and evaluating answers on a scale from 1 for most to 6 for least important influencing parameter.
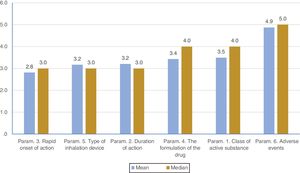
ResultsOverall, the most important parameters influencing patients... compliance according to physicians... perspective were rapid onset of action, type of inhalation device and duration of action. Adverse events were considered as the least important parameter. When COPD and asthma were examined separately, the most important parameters influencing compliance were rapid onset of action, ease of use and duration of action. Rapid onset of action was significantly more important in asthma than COPD.
ConclusionOnset and duration of action and ease of use were classified higher as important parameters to increase patients... compliance, according to physicians... perspective.
Obstructive airway diseases, Chronic Obstructive Pulmonary Disease (COPD) and Asthma, are increasing in frequency throughout the world, having a significant impact on quality of life. More than 3 million people died of COPD in 2012 accounting for 6% of all deaths globally.1,2
Similarly, the global prevalence rates of asthma in adults is increasing.3 Compliance plays a major role in disease outcome, as it has been associated with increased morbidity and mortality.4,5 A better understanding of the factors associated with adherence, can lead to more appropriate decisions regarding management. It has been shown that treatment success is proportionally linked to patients... compliance, both in COPD and asthma.6 On the other hand, suboptimal adherence to inhaled medication is linked to increased hospitalizations and economic health costs.7...10
The present study surveyed doctors... perspective on the role of active substance, type of inhalation device, onset of action, drug formulation, duration of action and adverse events as parameters influencing patients... compliance.
MethodsParticipantsA total of 144 prescribing doctors participated in the study with the majority being pulmonologists. Specifically, there were 104 pulmonologists (72%), 3 intensivists, 7 allergologists, 27 internists & 3 pediatricians) participated in the study. Excluding the 3 pediatricians, all other participants declared that were actively involved in the management of COPD and asthma. Regarding the workplace of respondents, 74% worked in a public hospital, 19% in a private practice and 7% in a private clinic.
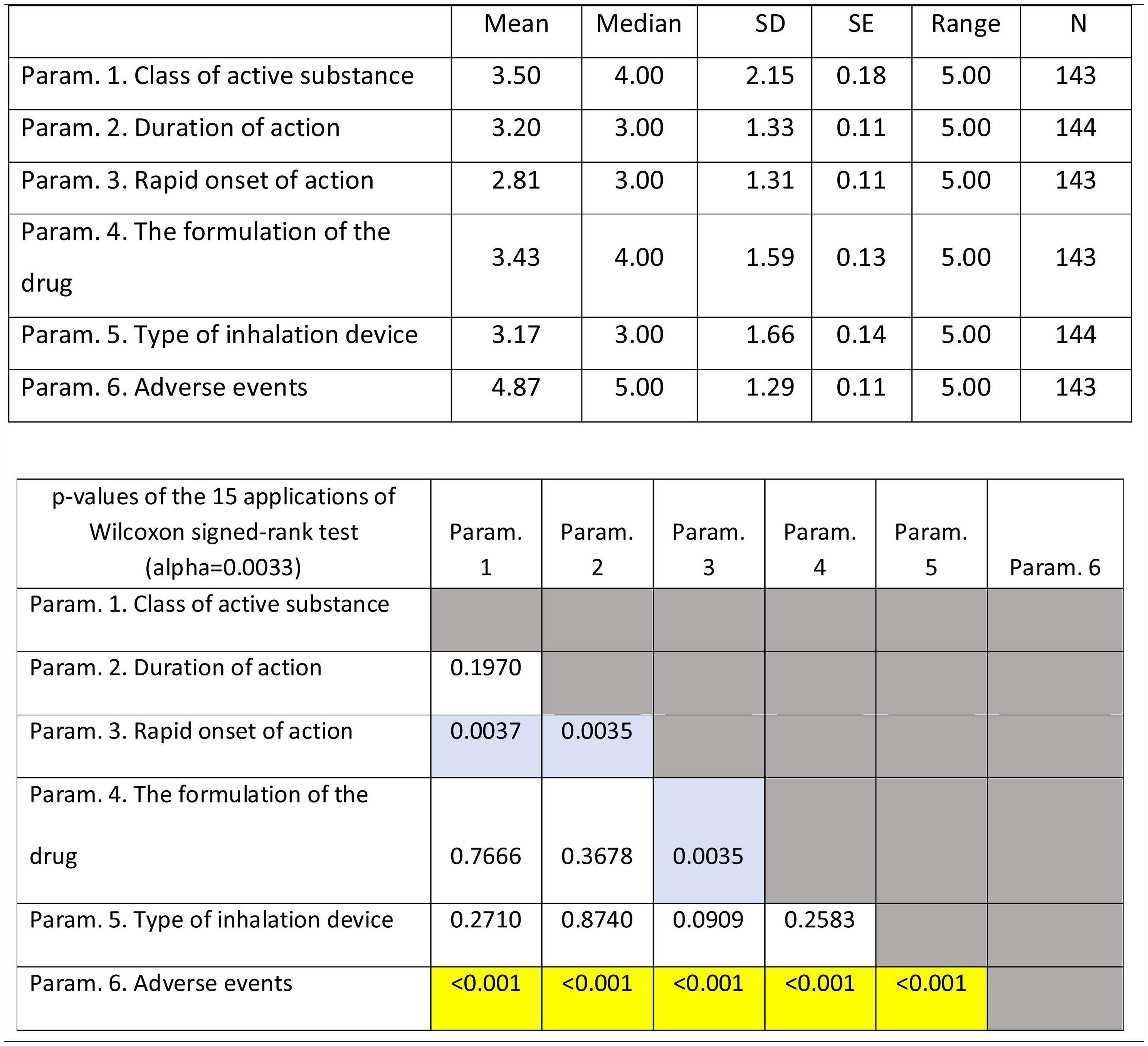
Design of the studyDuring the National Respiratory Congress, physicians were asked to answer a questionnaire regarding the importance of device and substance characteristics influencing the compliance of patients with chronic obstructive airways diseases. The importance of parameters was evaluated on a scale of 1...6, with one representing the most and six representing the least important. Evaluated parameters included rapid onset of action, duration of action, ease of use, low flow resistance, ability of the device to confirm the dose, type of inhalation device, and the formulation of the drug. Participants were asked to determine the importance of the above-mentioned parameters in chronic obstructive lung diseases in general and subsequently in COPD and asthma separately. The average time to complete the questionnaire was 12...min.
Statistical analysisDescriptive statistics: The continuous (e.g. age) and ordinal study parameters (e.g. the scaled answers to the Study questions) were presented using mean, median, standard deviation (SD), standard error of the mean (SE) and range, while the nominal parameters (e.g. gender) were presented using tables of frequencies.
Inference: The data analysed were subjective rankings (scores) by the physicians, on various parameters (characteristics) of inhalation devices. In the case of Question 1 (overall evaluation of parameters influencing patients... compliance/ 6 parameters) the data were ranked on a six-point scale. In the case of Question 2A (parameters influencing the compliance of patients with Asthma / 7 parameters) and Question 2B (parameters influencing COPD patients... compliance / 7 parameters) the data were ranked on a seven-point scale.
All above parameters were compared between male and female physicians using Mann-Whitney U-test, while they were compared between age groups using Kruskal Wallis test.
For the parameters belonging to Question 1 (overall evaluation of parameters influencing patients... compliance /6 parameters), Friedman...s test checked the overall difference between scores, i.e. if at least one of the parameters had a different score from the rest. Afterwards these six parameters were compared with each other using Wilcoxon signed-rank test (15 comparisons). Bonferroni correction was also applied, so the alpha level (two-tailed) for these comparisons was set at 0.05/15...=...0.0033.
For the (scaled) parameters belonging to Question 2A (parameters influencing the compliance of patients with Asthma / 7 parameters), Friedman...s test checked the overall difference between scores, i.e. if at least one of the parameters had a different score from the rest. Afterwards the seven parameters were compared with each other using Wilcoxon signed-rank test (21 comparisons). Again, Bonferroni correction was applied, so the alpha level (two-tailed) for these comparisons was set at 0.05/21...=...0.0024. The same procedure as above was applied (the alpha level [two-tailed] for these comparisons was set at 0.05/21...=...0.0024) for the scaled parameters belonging to Question 2B (parameters influencing the compliance of patients with COPD / 7 parameters). Finally, the parameters influencing COPD and Asthmatic patients... compliance were compared between patients with Asthma (Question 2A) and COPD patients (Question 2B) using Wilcoxon signed-rank test (7 comparisons). No Bonferroni adjustment was applied since these comparisons were independent. All analyses were performed using R 4.0.3.
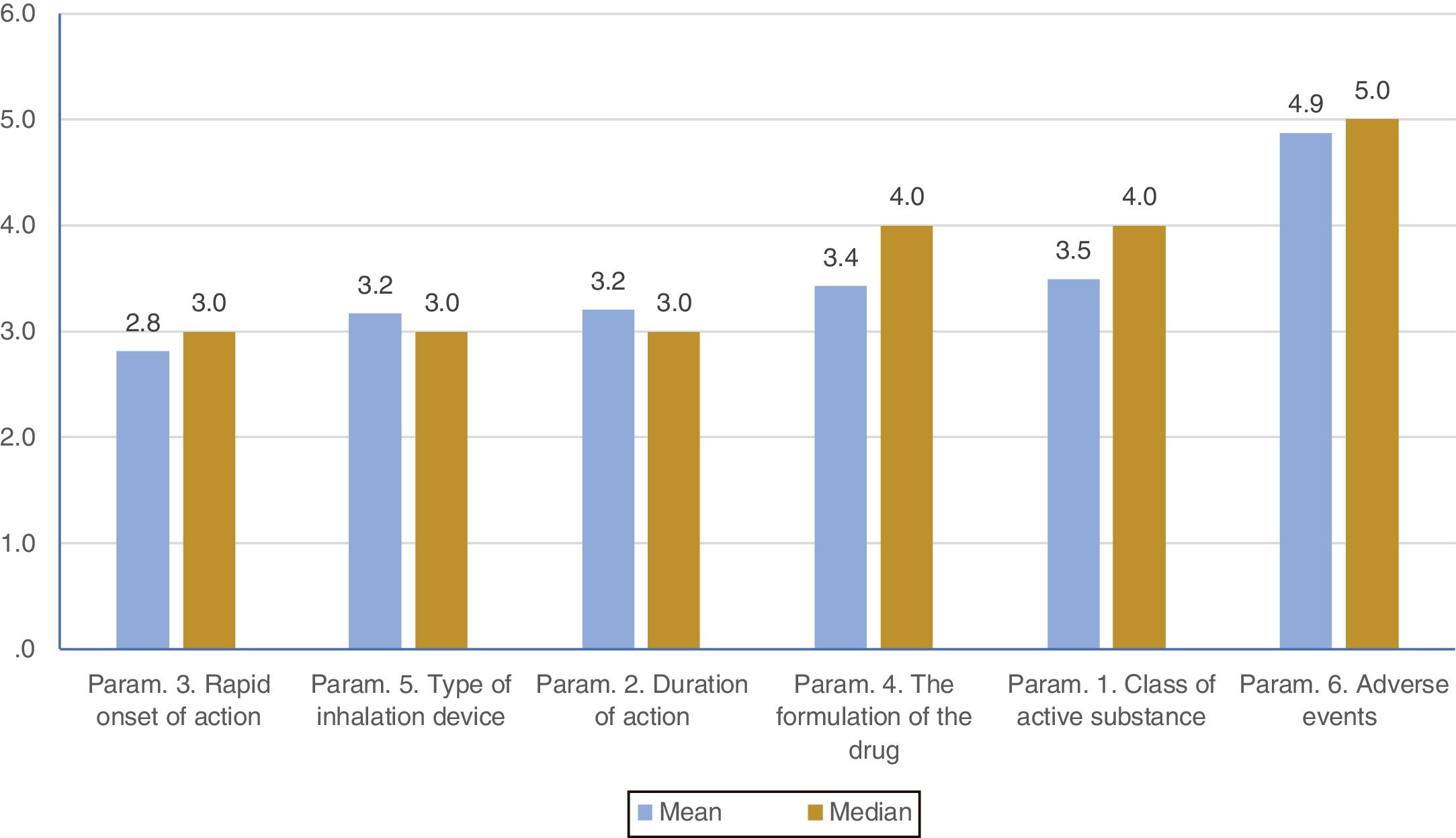
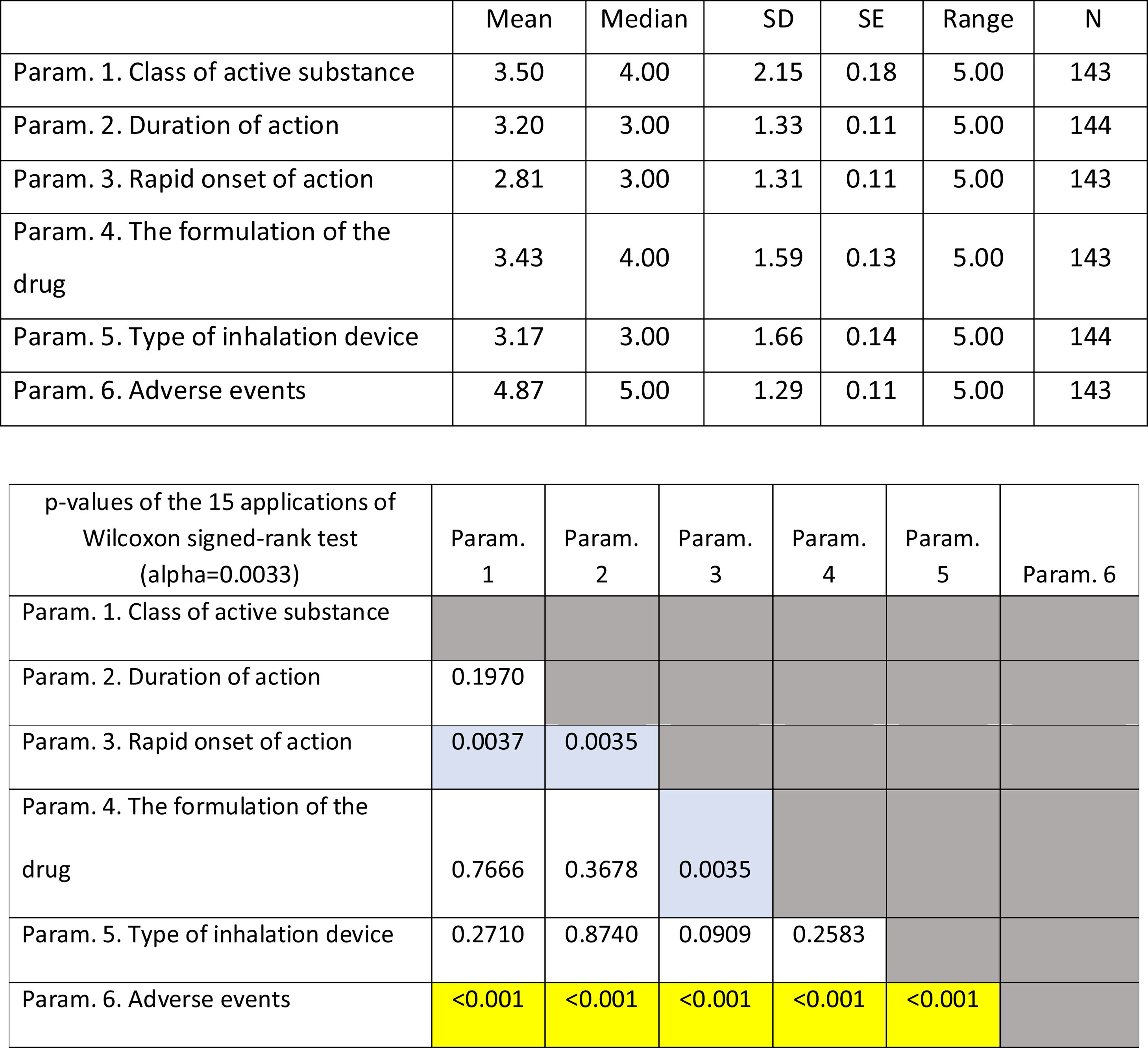
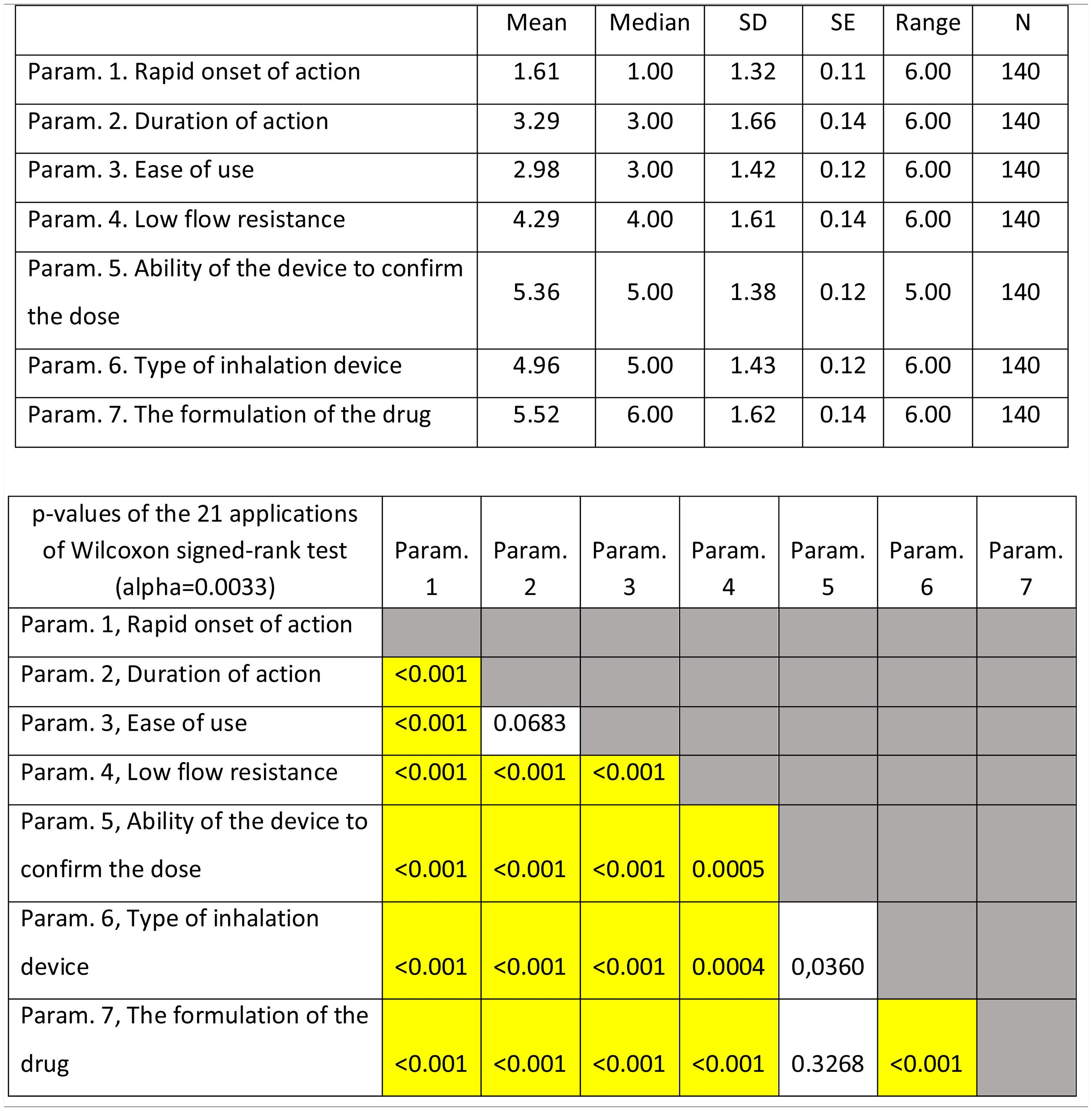
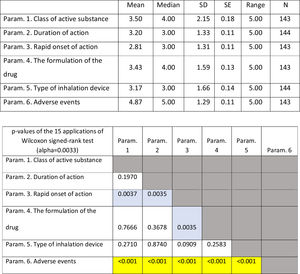
ResultsOverall evaluation of parameters influencing patients... complianceOn average ..úRapid onset of action..Ñ was ranked lower (more important- influential) than all the other parameters. It was statistically significantly lower than all other parameters except that of ..úType of inhalation device..Ñ. On the contrary, ..úAdverse events..Ñ was ranked statistically significantly higher (less important- influential) than all the other parameters. (Fig. 1). Statistical significance among studied parameters is shown in Table 1.
Descriptive statistics regarding the six Parameters of Question 1: Overall evaluation of parameters influencing patients... compliance. Scale: 1...=...1st most important, 6...=...6th least important. P-values of the 15 applications of Wilcoxon signed-rank test. Base: Observed cases.
Note: P-values that were marginally ...0.0033 are marked with blue colour, while p-values <0.0033 (Statistically significant difference between the two parameters) are marked with yellow colour.
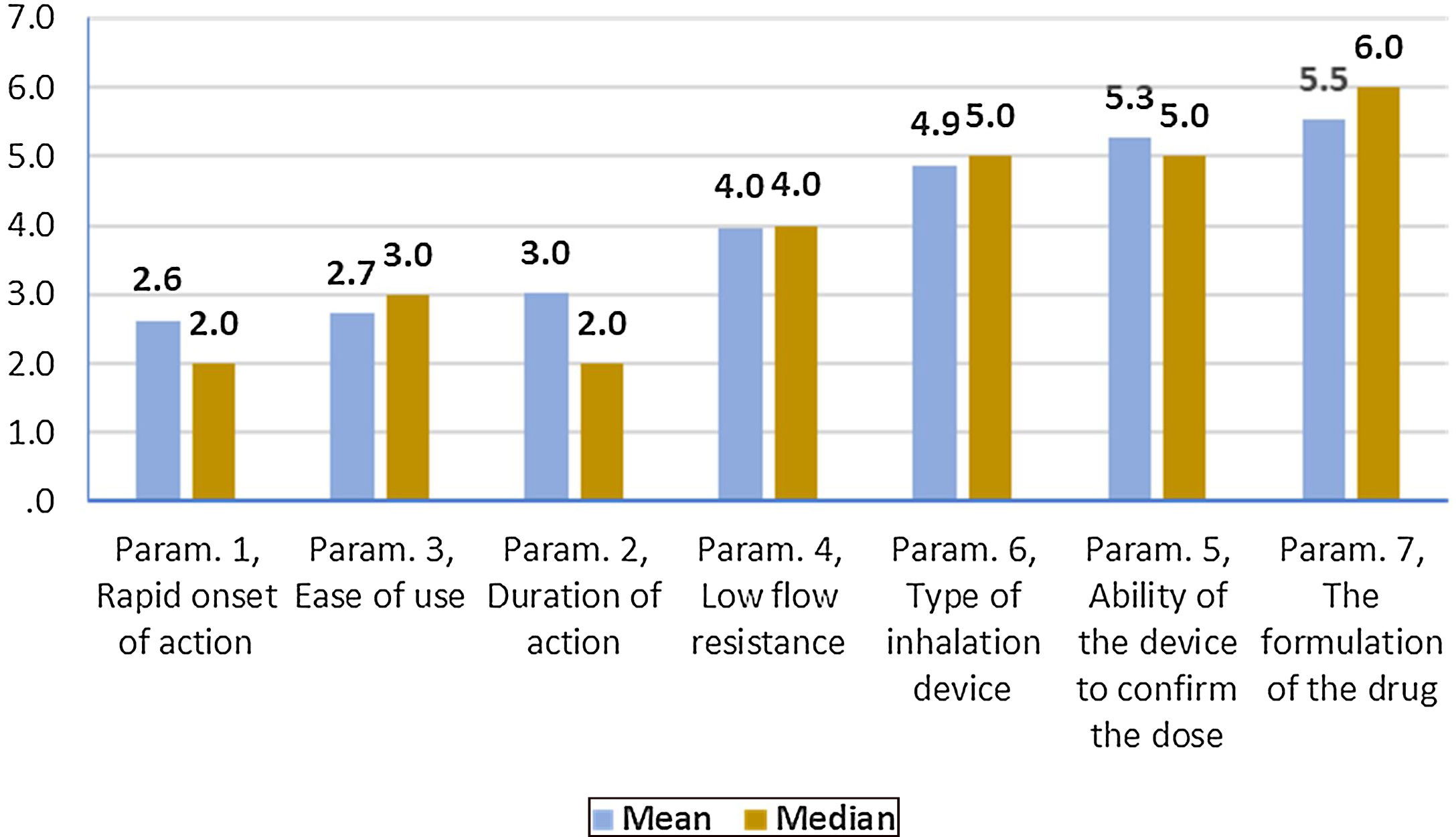
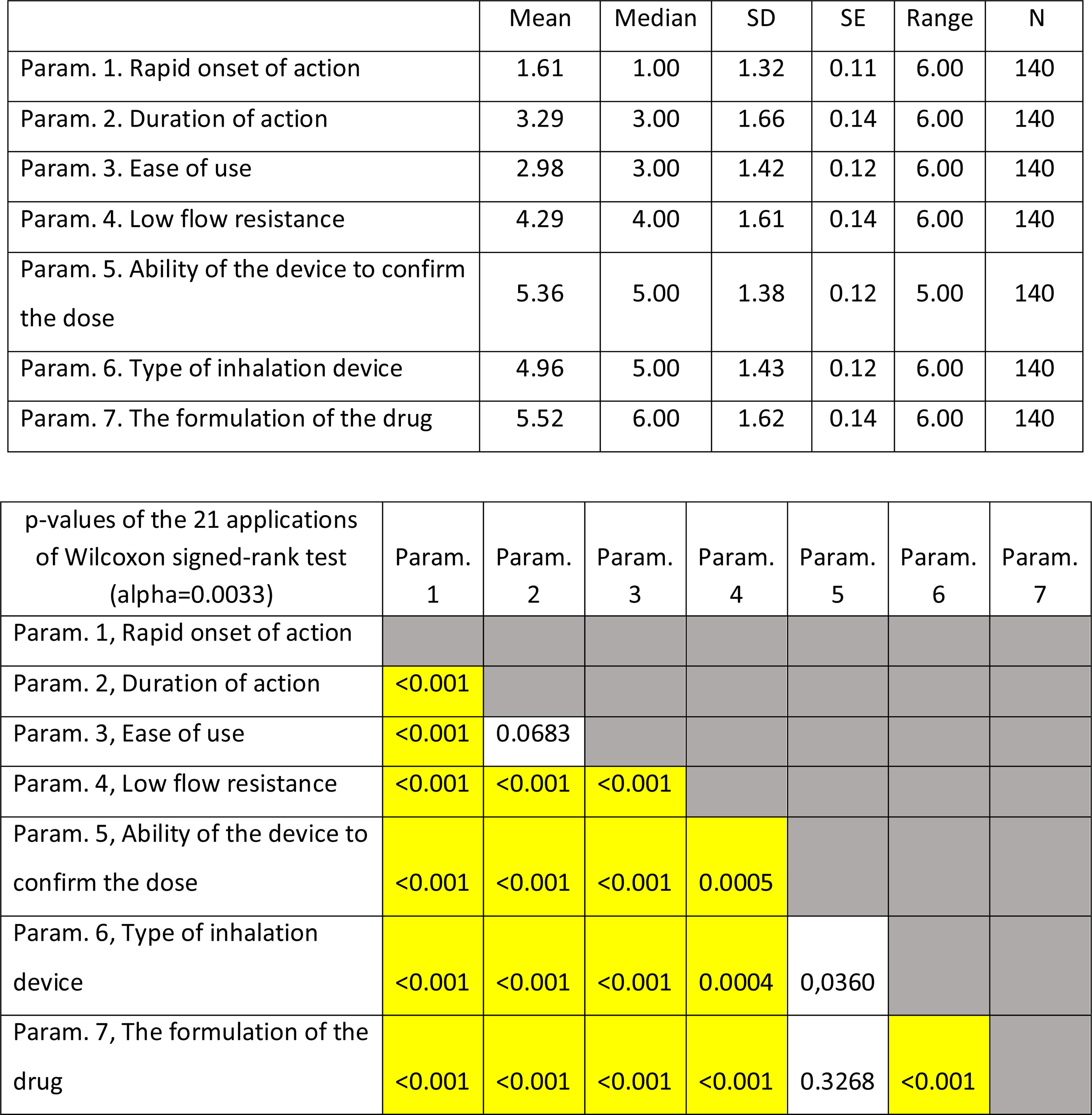
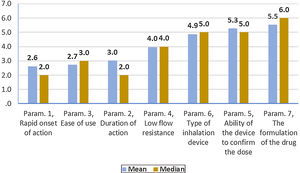
On average ..úRapid onset of action..Ñ was ranked statistically significantly lower (more important-influential) than all the other parameters. The next most influential parameters were ..úEase of use..Ñ and ..úDuration of action..Ñ that were ranked statistically significantly lower (more important- influential) than the other 4 parameters. On the contrary, ..úThe formulation of the drug..Ñ was ranked statistically significantly higher (less important) than all other parameters. (Fig. 2). Statistical significance among studied parameters is shown in Table 2.
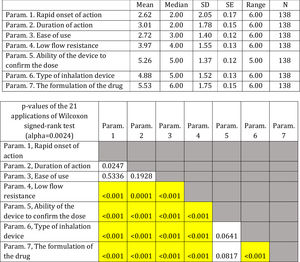
Descriptive statistics regarding the seven Parameters influencing the compliance of patients with Asthma. Scale: 1...=...1st most important, 7...=...7th least important. P-values of the 21 applications of Wilcoxon signed-rank test. Base: Observed cases (N...=...140).
Note: P-values<0.0024 (Statistically significant difference between the two parameters) are marked with yellow colour.
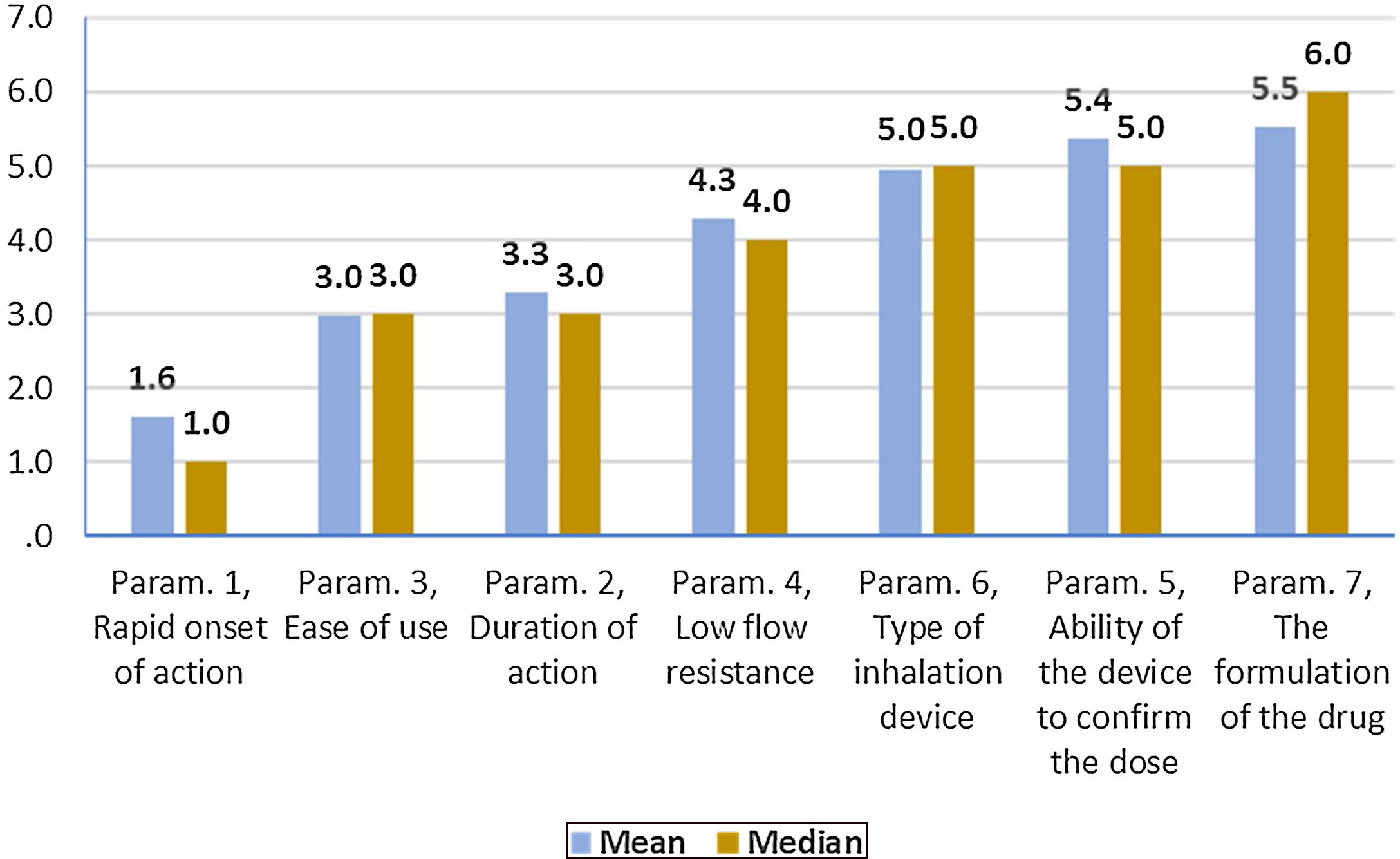
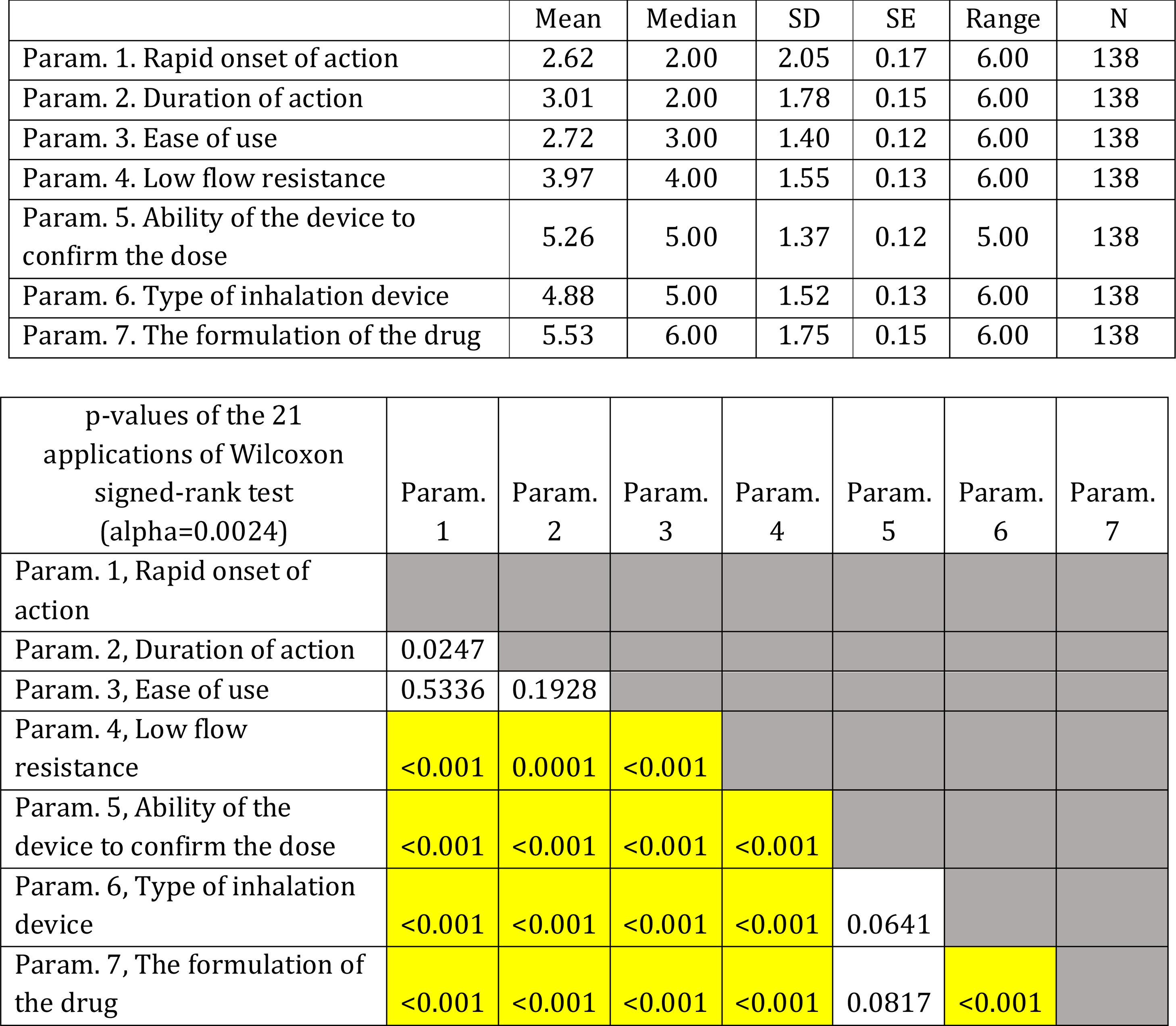
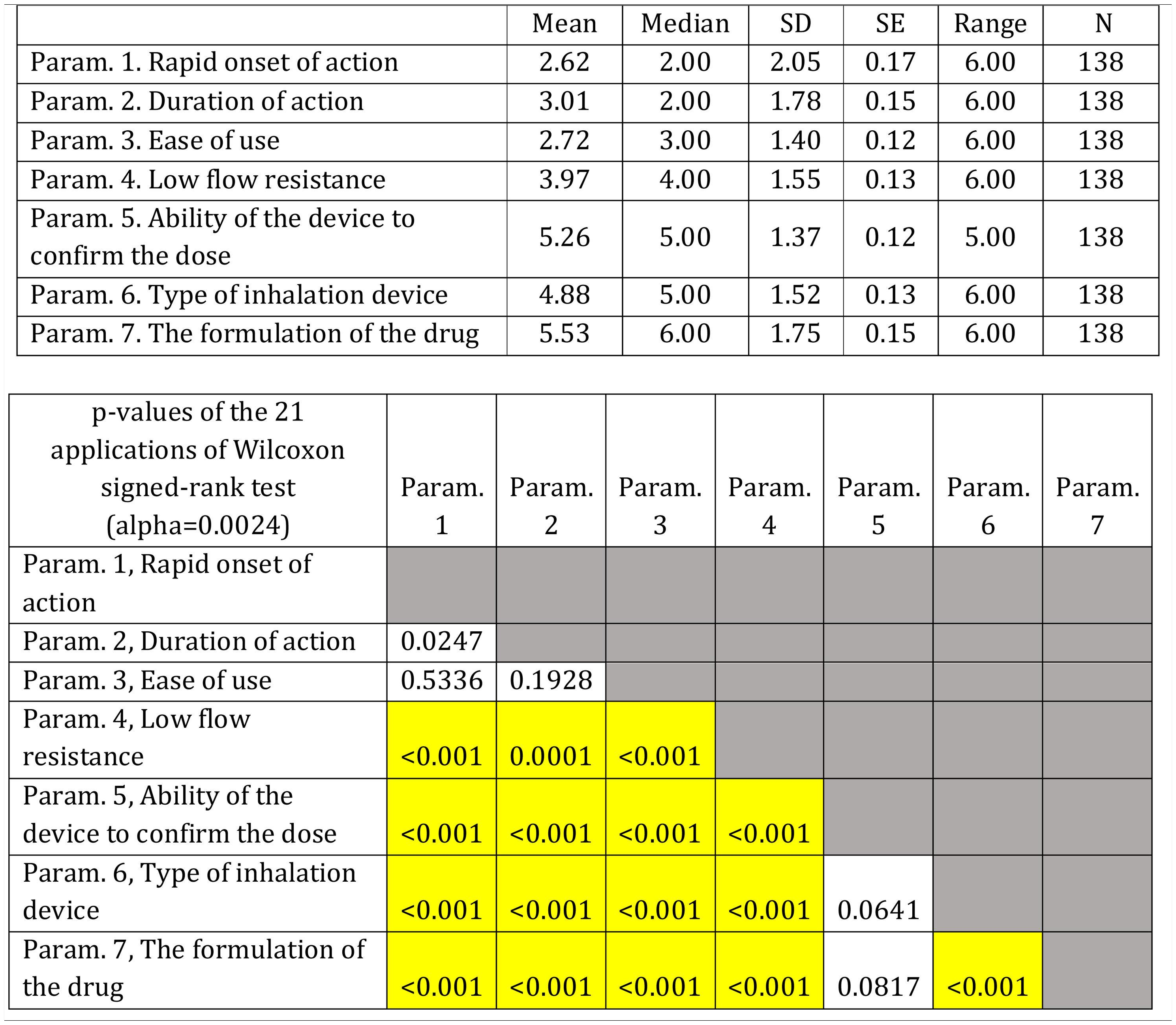
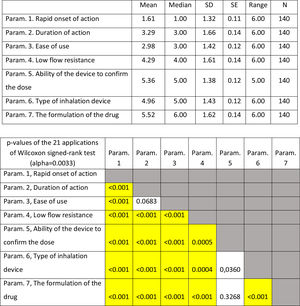
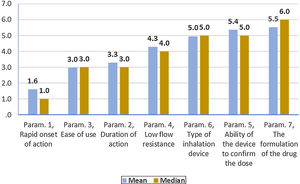
On average ..úRapid onset of action..Ñ, ..úDuration of action..Ñ and ..úEase of use..Ñ were ranked statistically significantly lower (more important-influential) than the other four parameters (..úRapid onset of action..Ñ, ..úDuration of action..Ñ and ..úEase of use..Ñ were not statistically significantly differentiated between each other). The parameter ..úThe formulation of the drug..Ñ was ranked statistically significantly higher (less important) than all other parameters, except that of ..úAbility of the device to confirm the dose..Ñ.
(Fig. 3). Statistical significance among studied parameters is shown in Table 3.
Descriptive statistics regarding the seven parameters influencing the compliance of patients with COPD. Scale: 1...=...1st most important, 7...=...7th least important. P-values of the 21 applications of Wilcoxon signed-rank test. Base: Observed cases (N...=...138).
Note: P-values<0.0024 (Statistically significant difference between the two parameters) are marked with yellow colour.
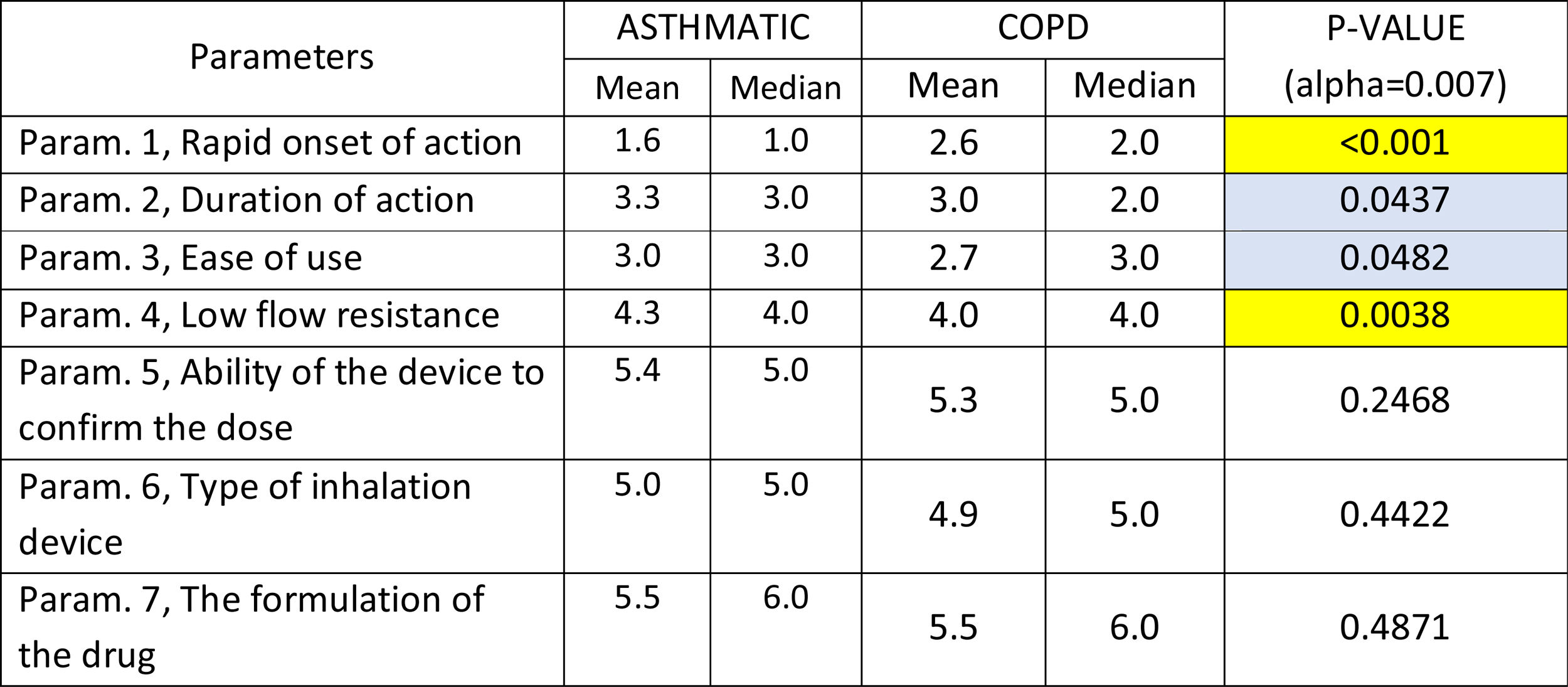
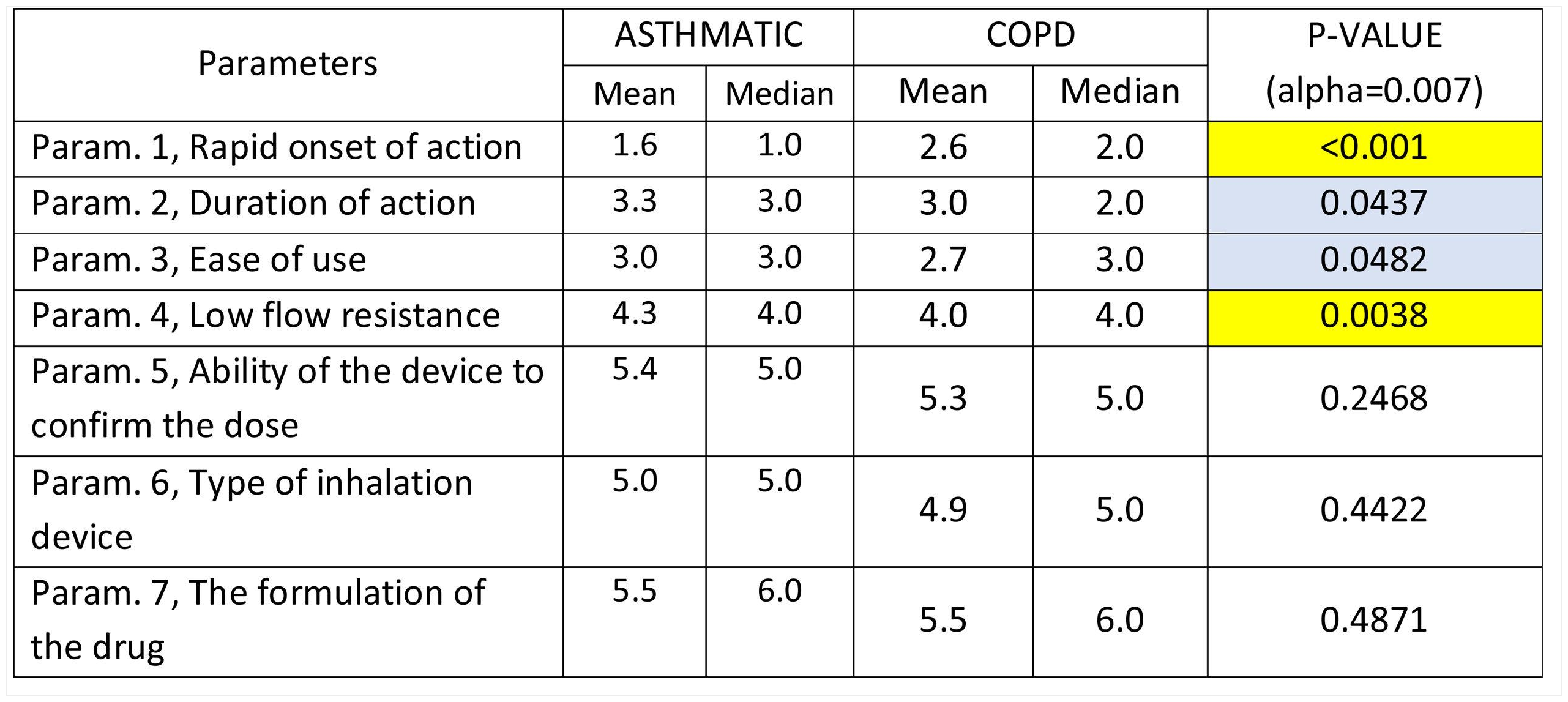
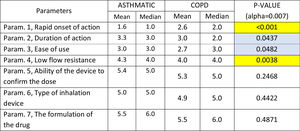
The following parameters were differentiated in the comparison between patients with Asthma and COPD patients,: Rapid onset of action was ranked statistically significantly lower (more important-influential) in Asthmatic patients, Low flow resistance was ranked statistically significantly higher (less important-influential) in Asthmatic patients, duration of action and ease of use were marginally statistically significantly higher (less important-influential) in Asthmatic patients (Table 4).
Mean and Median values regarding the seven Parameters influencing the compliance of patients with ASTHMA/ COPD. P-values of the 7 applications of Wilcoxon signed-rank test. Base: Observed cases (N...=...138).
Note: P-values marginally <0.05 are marked with blue colour, while p-values <0.005 are marked with yellow colour.
In this study surveying the opinion of physicians treating patients with chronic obstructive airways diseases (asthma and COPD), rapid onset of action, ease of use and duration of action were the most important parameters favoring patients... compliance for both diseases.
As expected, rapid onset of action was the most important factor influencing compliance both in COPD and asthma. Interestingly, it was statistically more important in asthma than in COPD. This finding reflects the difference in pathophysiology and clinical behavior between asthma in COPD as perceived by physicians. In general, asthma is considered a disease which is more likely to be complicated with episodes of acute shortness of breath in need of immediate relief. Duration of action was also considered as an important parameter influencing compliance, irrespective of the underlying diagnosis. This parameter was numerically more important in COPD compared to asthma. Duration of action is clinically relevant for bronchodilators as their duration of action has a direct impact on patients... symptoms and therefore compliance. The above physicians views also correlate with patients... perspectives.11 The fact that adverse events were not considered at all as an important compliance factor, confirms the strong belief of physicians in the safety of inhaled medications. Ease of use was also considered as a key factor for compliance. In asthma ease of use is especially important during an acute event of dyspnea needing immediate relief. In COPD patients, who are usually older with several comorbidities ease of use is also an understandable parameter that can affect compliance.
The increased number of inhaler devices in recent years has resulted in a confusing number of choices for physicians and patients. The prescribing physician must balance the efficacy of inhaled medications with patients... preferences, personality traits and their physical ability to handle devices appropriately. The selection of an inhalation device for patients with asthma and COPD has become a complicated issue as several parameters should be taken into account, such as: device/drug availability, clinical setting, patient age, the ability to use the selected device correctly, device use with multiple medications, cost and reimbursement, drug administration time, convenience in both outpatient and inpatient settings, and physician and patient preference, as stated previously.12...16 Unfortunately, such studies are scarce in the literature.
Strengths of this study are the detailed questionnaire, and the relatively high number of respondents. However, we acknowledge certain limitations of the study. First, we recognize that we explored a complex issue which is difficult to approach by "one- shot strong and direct answers..Ñ as the answers do not take into account the individualization of the patient path, the level of functional severity, the different phenotypes, the age of the patient and the patient's history. Another limitation is that this study exclusively included specialists and lacks the views of general practitioners. Thus, the results cannot be generalized for non-specialists. Furthermore, the study reflects the beliefs of the physicians of a single country and comparative studies among different countries could provide significant insights. Given the limited number of relevant studies, the present work adds to our understanding of physicians... perspectives on patient compliance.
In conclusion, rapid onset of action, ease of use and duration of action, were classified higher among the parameters examined. Further studies are needed among physicians of different specialties and nationalities to identify more rigorously factors increasing patient compliance. Evidence-based guidelines for the selection of the appropriate inhalation device in specific clinical settings are needed.
Conflict of interestAll authors have no conflicts of interest to disclose.