Chronic cough (CC) is a common disease that has a detrimental impact on quality of life. In everyday clinical practice, one of the main challenges of CC is its aetiological work-up. Indeed, CC is associated with a high number of potential causes including asthma, gastro-oesophageal reflux disease (GERD) and rhinosinusitis. Over the past 15 years, a number of case reports1,2 and studies with retrospective design and/or small sample sizes3-5 have indicated that obstructive sleep apnoea (OSA) could also trigger CC. A larger prospective study is, however, needed to establish whether OSA should be considered a potential cause of CC.
The aim of this study was, therefore, to investigate the association between CC and moderate to severe OSA in a large prospective cohort of patients investigated for suspected OSA.
Consecutive patients undergoing in-laboratory polysomnography [PSG] or type-3 home sleep apnoea testing for clinical suspicion of OSA were prospectively recruited in two French hospitals. All participants gave an informed consent for their participation in the study. This study was approved by the University of Angers ethics committee (Comité d'Ethique du Centre Hospitalier Universitaire d'Angers, No. 2007/17; Comité Consultative sur le Traitement de l'Information en matière de Recherche dans le domaine de la Santé, 07.207bis). Data regarding clinical characteristics and metrics of severity derived from sleep recordings were collected. CC was defined as a cough that persisted for at least 8 weeks.6,7
Moderate to severe OSA was defined as Apnoea/Hypopnoea Index (AHI) ≥15/h. Respiratory events were scored manually using the following criteria: apnoea was defined as an at least a 90 % decrease in the oronasal sensor signal; while hypopnoea was defined as an at least a 30 % decrease in the nasal pressure signal combined with either >3 % arterial oxygen desaturation or arousal (PSG), both lasting at least 10 s.8 The cough itself was evaluated using the Leicester Cough Questionnaire (LCQ) and the Hull Airway Reflux Questionnaire (HARQ).
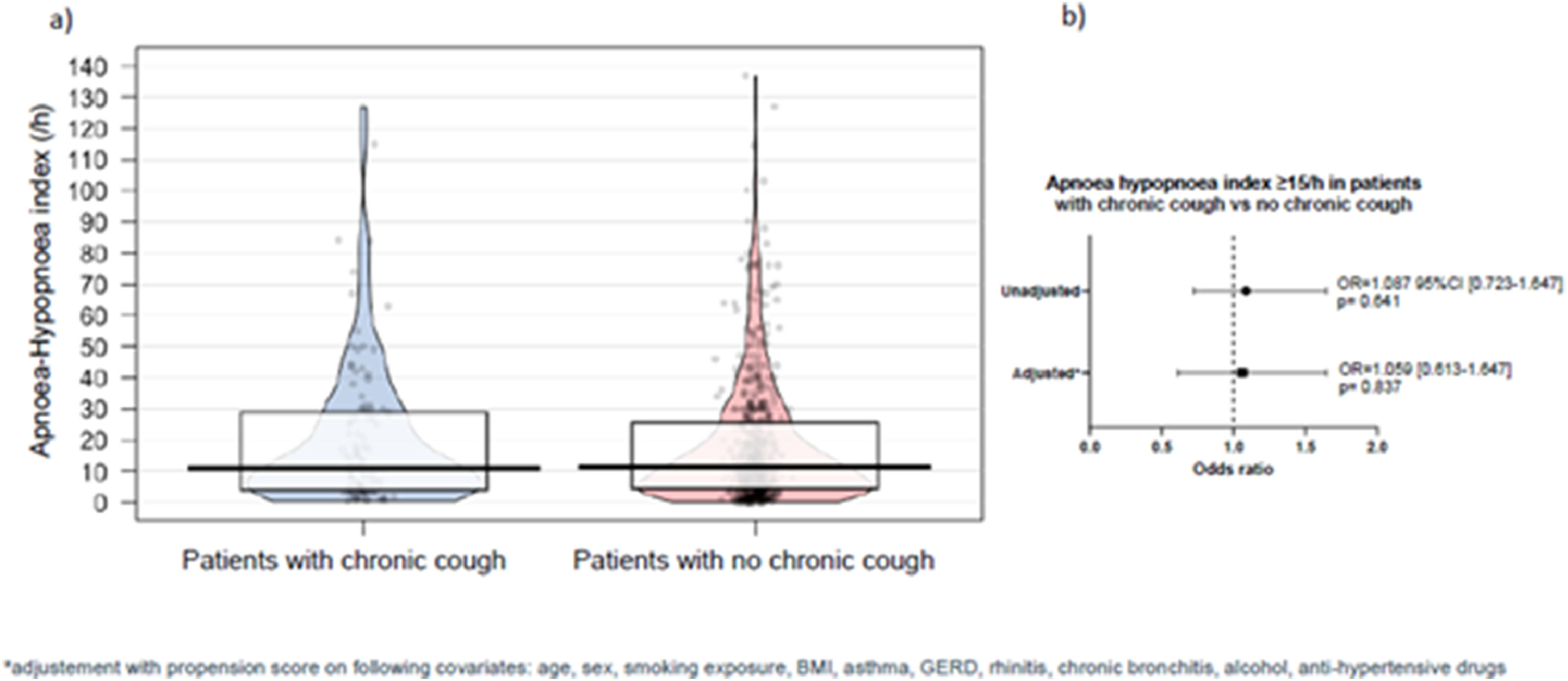
A number of studies have been conducted to date that examine the prevalence of CC in patients with OSA. According to the findings of these studies, the proportion of patients with CC among those with moderate to severe OSA (AHI≥15/h) is 39 %.4,5 Given that this percentage is likely to be overestimated, we assume that the percentage of OSA patients with CC is more likely to be around 20 %, compared with 12 % in patients without this condition.9 In order to meet this study's primary objective, which is to demonstrate that the rate of CC among OSA patients is distinct from its occurrence in other conditions, it is necessary to include 752 patients to guarantee a power of 80 % while controlling the risk of first kind at the 5 % threshold. Statistical analysis was performed using R software (version 3.5.1, www.R-project.org, Vienna, Austria). Data were compared using Fisher's exact test for categorical variables and the Mann-Whitney U test for continuous variables. The violin plot was used to compare the distribution of AHI values between patients with and without CC (unadjusted population). A matching propensity score was used with an average treatment effect (ATM) estimand to reduce the bias due to confounding variables. The following covariates were adjusted using propensity scores: age; sex; smoking exposure; BMI; asthma; GERD; rhinitis; chronic bronchitis; alcohol use; use of anti-hypertensive drugs.
In total, 1052 patients were enrolled in the study. Due to technical issues with particular sleep recordings or missing data, 821 patients were included in the final analysis. Of these patients, 44.3 % were female, the mean age was a mean age of 52.9 ± 14.9 years and the mean BMI of 30.18±7.0 kg/m². The prevalence of CC in the whole population was 13.4 %. The mean AHI was 20.1 ± 22.0/h. Severe OSA (AHI≥30/h) was observed in 206 (25.1 %) patients and moderate to severe OSA (AHI ≥15/h) in 360 (43.9 %) patients. As expected, patients with CC compared to those with no CC were more likely to present chronic bronchitis (23.6 % versus 6.3 % respectively; p < 0.001) and GERD (53.6 % versus 31.5 % respectively; p < 0.001) as compared to the no CC group. No further differences were observed regarding age, gender, BMI, smoking status and the prevalence of diabetes, hypertension and asthma. In the subgroup of patients investigated with polysomnography, no differences were observed between the groups in terms of total sleep time, arousal index and sleep structure (light, deep and REM sleep percentage).
According to the unadjusted analysis, no differences were identified between patients with and without CC in terms of AHI values (Fig. 1a) and AHI categories (p = 0.641). After applying the propensity score (adjusted analysis), no association between CC and moderate to severe OSA (p = 0.837) was identified (Fig. 1b). Similar results were observed for mild, moderate and severe OSA. The prevalence of CC was also similar according to AHI categories: 14.8 % in patients with AHI<5/h, 12.6 % with AHI 5–15/h, 13.6 % with AHI 15–30/h and 12.6 % with AHI ≥30/h.
a) Comparison of Apnoea/Hypopnoea Index (AHI) values between patients with and without chronic cough (unadjusted population); b) Hazard ratio of AHI ≥ 15/h comparing patients with chronic cough to those without in the unadjusted population and with a propensity score including the following covariates: age, sex, smoking exposure, BMI, asthma, GERD, rhinitis, chronic bronchitis, alcohol and anti-hypertensive drugs. Boxes indicate the 75th percentile (top horizontal line), median (black bold horizontal line) and the 25th (bottom horizontal line) percentiles of the distribution, and a rotated kernel density plot surrounds the box (shaded area) on each side.
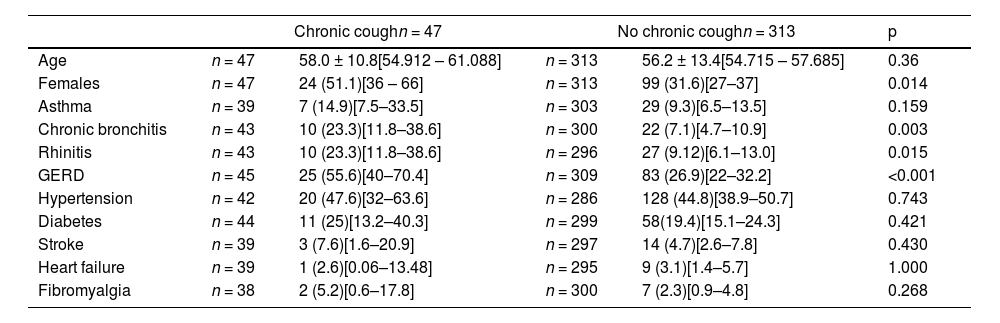
In order to better understand CC mechanisms in OSA patients, we compared the patients in the group of patients with moderate to severe OSA according to whether they had CC or not (Table 1). Interestingly, many of the comorbidities that trigger coughing were statistically more frequent in patients with OSA and CC compared to those with OSA but no CC. Indeed, among moderate to severe OSA, the proportion of patients with GERD (25 [55.6 %] vs. 83 [26.9 %]), chronic bronchitis (10 [23.2 %] vs. 27 [7.3 %]) and rhinitis (10 [23.2 %] vs. 27 [9.1 %]) was statistically higher in patients with CC than in those without. We found no differences, however, when comparing the prevalence of comorbidities between male and female patients with CC.
Comorbidities in moderate-to-severe OSA patients with and with no chronic cough. Data are expressed as mean±SD or number (%) with [95 % confident interval].
In order to determine whether patients with OSA and CC have a specific profile, we compared patients with CC and OSA (n = 47) and those with CC but no OSA (n = 63). We found no differences between the two groups in terms of GERD or cough severity (LCQ and HARQ scores).
To date, this is the largest prospective cohort study to research the link between OSA and CC. We found no differences in terms of AHI values between patients with and without CC. Similarly, there were no differences in CC prevalence according to AHI categories. It is noteworthy that the global prevalence of CC in our population was similar to that observed in a large meta-analysis.9 Above all, those data contradict previous positive observations that were based on limited sample sizes and led to European guidelines to consider OSA as a potential CC risk factor.10
A relatively high prevalence of OSA was found by Sundar et al. in their retrospective study involving 75 patients with CC.3 Their study did not, however, include a control group and their sleep recordings were performed solely in a limited number of patients, thus, making it difficult to draw any firm conclusions. A large survey study featuring 4217 participants showed that patients with CC (2.9 %) had significantly higher STOP-Bang scores than those without CC.11 As STOP-Bang includes BMI, age and sex, which are also considered to be CC risk factors, it is difficult to discern just how relevant this association may be without making further adjustments or diagnostic sleep recordings. In a retrospective study involving 131 patients referred to a sleep laboratory for suspected OSA, CC prevalence in the OSA group was significantly higher than in the non-OSA group (39.4% vs. 12.5 %).4 Quite in keeping with our results, however, the association between AHI and CC was no longer significant in the multivariable analysis.
A positive association between OSA and CC identified in previous studies is likely to be largely accounted for by confounding factors. In our study, propensity score matching was applied in order to guarantee a balance of covariates between each group. These covariates were age, sex, smoking exposure, BMI, asthma, GERD, rhinitis, chronic bronchitis, alcohol use and the use of anti-hypertensive drugs. In order to identify factors that explain the positive association between OSA and CC in other studies, we compared OSA patients with CC to those without CC (Table 1). We found that the proportion of patients with chronic bronchitis, rhinitis and GERD were significantly higher in those with CC compared to those with no CC. Asthma was also more frequently observed in patients with CC compared with those without it, but the difference in such cases was not significant, probably due to a lack of power. These findings highlight that CC is often multifactorial and tends to be triggered by the usual causes of a cough. Therefore, according to our results, any unique and strong association between CC and OSA is unlikely.
While GERD has traditionally been associated with OSA and CC,4 we found that a number of other comorbidities (asthma, rhinitis, stroke, etc.) offer likely explanations of why patients with OSA also suffer from CC. Due to a small number of patients for whom LCQ scores at 3 months following the initiation of continuous positive airway pressure (CPAP) treatment were available, we were not able to analyse the effects of CPAP treatment on cough. Notably, a recent large observational study demonstrated that while CPAP does indeed impact GERD severity, it failed to have a significant effect on daytime CC.12 As already suggested by previous studies, a nonspecific impact of CPAP on GERD, independently of the presence of OSA and its severity, cannot be excluded to explain those results.13
In line with other studies on CC, we measured cough severity using questionnaires (LCQ and HARQ). Yet the main limitation of such questionnaires is their lack of objectivity; indeed, using a cough monitor is likely to provide more accurate and objective data regarding the severity of a cough. That said, a significant correlation between objective cough frequency and LCQ has been demonstrated elsewhere.14
In conclusion, we found no association between CC and OSA in a large cohort of patients investigated for suspected OSA. Further studies are, however, needed in order to determine which patients with CC would benefit from being screened for OSA.