The 2009 swine flu pandemic caused by Influenza A virus subtype H1N1 (H1N1) virus affected more than 214 countries and overseas territories or communities and over 18,449 deaths caused by the H1N1 infection1 were confirmed. After ten years, a new pandemic named Coronavirus Disease 2019 (COVID-19) caused by severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) virus emerged in 2019 in the city of Wuhan, China. Both diseases were declared a pandemic by the World Health Organization (WHO), swine flu on 24th April 2009 and COVID-19 on 11th March 2020. To date, 7th July 2020, the COVID-19 disease affected nearly 12 million inhabitants reaching 213 countries and territories around the world and two international conveyances. In addition, ∼550,000 deaths were associated with the disease in 185 locations worldwide. The COVID-19 started in China and spread worldwide changing its epicenter first to Europe, followed by the United State of America and, now, from May to July 2020 South America, mainly affecting patients in Brazil causing the health system to collapse in several states such as Amazonas and Rio de Janeiro. On 7th July 2020, Brazil presented a total number of 1,674,655 patients with COVID-19; 535,558 active cases and 1,072,229 clinically recovered cases, 66,868 deaths related to the disease, and a case fatality rate of 3.99. Brazil occupies the 105 position worldwide for the number of real-time polymerase chain reaction by one million of inhabitants to screen SARS-CoV-2 virus.2,3
Both the 2009 swine flu pandemic and 2019/2020 COVID-19 pandemic resulted in a high number of published articles in a short period of time. The number of publications can be associated with the great impact on science in several areas including medicine, sociology, environment, physics, mathematics, biology and many other knowledge areas that act individually or are multidisciplinary in scope. Another striking aspect is that the collection of articles and other resources on the COVID-19 outbreak, including clinical reports, management guidelines, and comments are freely available to be used by researchers, health professionals and the community. The free availability of data resulted from the commitment of top scientific (e.g. Science and Nature) and Medical (e.g. New England Journal of Medicine and Lancet) journals to supply online articles fast and free of charge. To date, the Public Health Emergency COVID-19 Initiative has over fifty authors publishing (collaborators) that have volunteered to make their coronavirus-related articles accessible in PubMed Central® in formats and under license terms that facilitate text mining and secondary analysis.
In our study, a comparison was made between both pandemics regarding the number of publications divided into six main themes as follows: (i) the number of articles related with each disease caused by H1N1 virus and SARS-CoV-2 virus; (ii) the number of articles related with each disease considering the theme diagnosis; (iii) the number of articles related with each disease considering the theme symptoms; (iv) the number of articles related with each disease considering the theme epidemiology; (v) the number of articles related with each disease considering the theme treatment; and (vi) the number of articles related with each disease considering the theme vaccine. Each theme was individually analyzed and no exclusion was carried out using a specific theme as dominant because each theme cooperated equally to improve the Scientific knowledge during both pandemics. Moreover, one article can contain information about two (or more) themes with equal contextualization.
The data search was done using the PubMed (Public/Publisher MEDLINE) (https://pubmed.ncbi.nlm.nih.gov) using of the following descriptors: (i) (swine flu OR H1N1 OR H1N1 influenza virus OR influenza virus OR H1N1 influenza OR H1N1/09 OR H1N1 virus OR A(H1N1)pdm09 OR H1N1 flu OR Mexican flu OR influenza A); (ii) (coronavirus disease-19 OR coronavirus disease OR corona virus OR COVID-19 OR COVID19 OR SARS-CoV-2); (iii) the descriptors of (i) and (ii) were used along with the following descriptors: (iiia) AND (diagnosis); (iiib) AND (symptoms); (iiic) AND (epidemiology); (iiid) AND (treatment); (iiie) AND (vaccine). The data extraction was carried out weekly for ten weeks after the WHO declared the swine flu (25th April 2009) and COVID-19 (11th March 2020) as pandemic (Table 1 shows the periods of analysis).
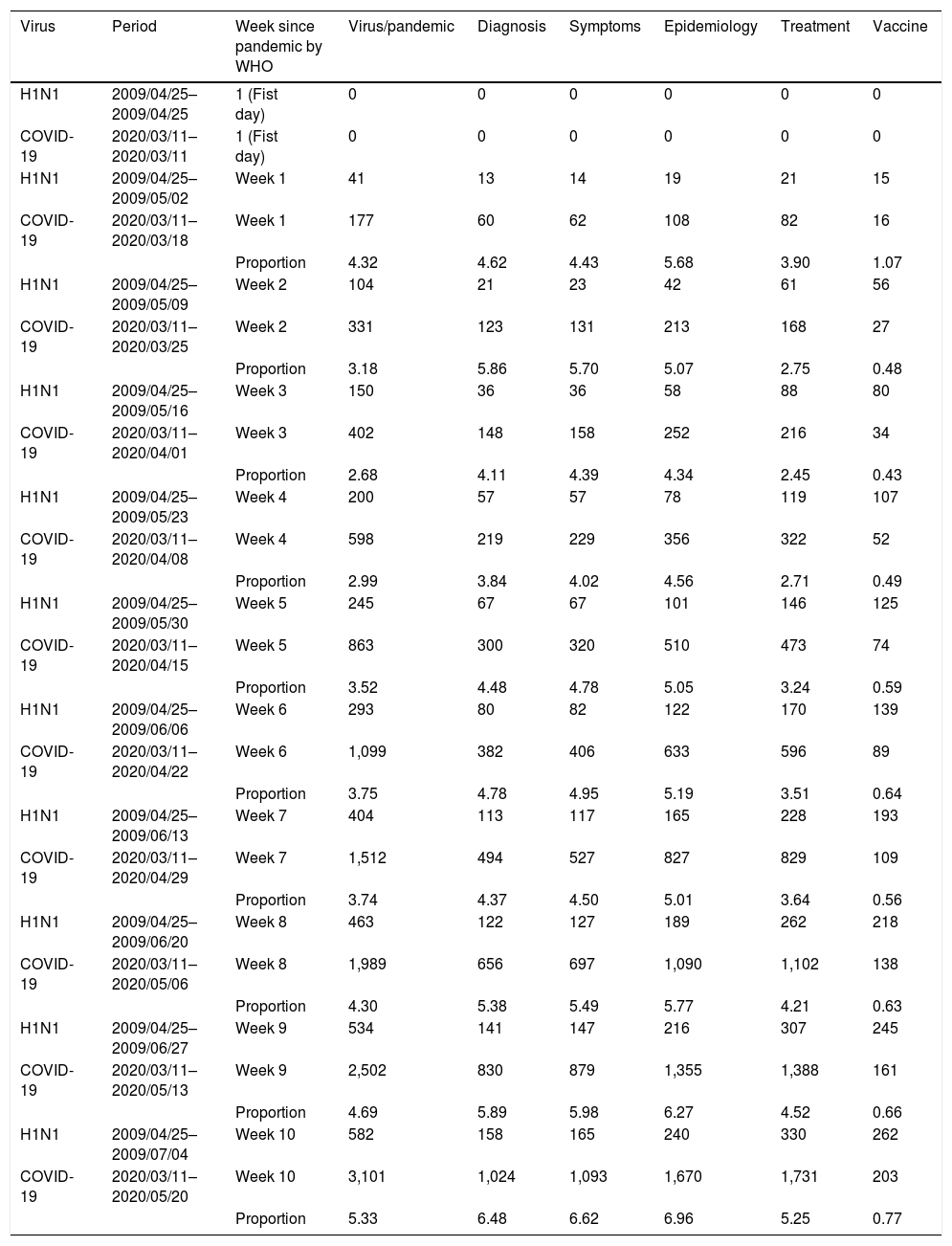
Number of publications after the World Health Organization (WHO) declared swine flu disease (2009) and COVID-19 (2020) disease as pandemics distributed in a period of ten weeks.
| Virus | Period | Week since pandemic by WHO | Virus/pandemic | Diagnosis | Symptoms | Epidemiology | Treatment | Vaccine |
|---|---|---|---|---|---|---|---|---|
| H1N1 | 2009/04/25–2009/04/25 | 1 (Fist day) | 0 | 0 | 0 | 0 | 0 | 0 |
| COVID-19 | 2020/03/11–2020/03/11 | 1 (Fist day) | 0 | 0 | 0 | 0 | 0 | 0 |
| H1N1 | 2009/04/25–2009/05/02 | Week 1 | 41 | 13 | 14 | 19 | 21 | 15 |
| COVID-19 | 2020/03/11–2020/03/18 | Week 1 | 177 | 60 | 62 | 108 | 82 | 16 |
| Proportion | 4.32 | 4.62 | 4.43 | 5.68 | 3.90 | 1.07 | ||
| H1N1 | 2009/04/25–2009/05/09 | Week 2 | 104 | 21 | 23 | 42 | 61 | 56 |
| COVID-19 | 2020/03/11–2020/03/25 | Week 2 | 331 | 123 | 131 | 213 | 168 | 27 |
| Proportion | 3.18 | 5.86 | 5.70 | 5.07 | 2.75 | 0.48 | ||
| H1N1 | 2009/04/25–2009/05/16 | Week 3 | 150 | 36 | 36 | 58 | 88 | 80 |
| COVID-19 | 2020/03/11–2020/04/01 | Week 3 | 402 | 148 | 158 | 252 | 216 | 34 |
| Proportion | 2.68 | 4.11 | 4.39 | 4.34 | 2.45 | 0.43 | ||
| H1N1 | 2009/04/25–2009/05/23 | Week 4 | 200 | 57 | 57 | 78 | 119 | 107 |
| COVID-19 | 2020/03/11–2020/04/08 | Week 4 | 598 | 219 | 229 | 356 | 322 | 52 |
| Proportion | 2.99 | 3.84 | 4.02 | 4.56 | 2.71 | 0.49 | ||
| H1N1 | 2009/04/25–2009/05/30 | Week 5 | 245 | 67 | 67 | 101 | 146 | 125 |
| COVID-19 | 2020/03/11–2020/04/15 | Week 5 | 863 | 300 | 320 | 510 | 473 | 74 |
| Proportion | 3.52 | 4.48 | 4.78 | 5.05 | 3.24 | 0.59 | ||
| H1N1 | 2009/04/25–2009/06/06 | Week 6 | 293 | 80 | 82 | 122 | 170 | 139 |
| COVID-19 | 2020/03/11–2020/04/22 | Week 6 | 1,099 | 382 | 406 | 633 | 596 | 89 |
| Proportion | 3.75 | 4.78 | 4.95 | 5.19 | 3.51 | 0.64 | ||
| H1N1 | 2009/04/25–2009/06/13 | Week 7 | 404 | 113 | 117 | 165 | 228 | 193 |
| COVID-19 | 2020/03/11–2020/04/29 | Week 7 | 1,512 | 494 | 527 | 827 | 829 | 109 |
| Proportion | 3.74 | 4.37 | 4.50 | 5.01 | 3.64 | 0.56 | ||
| H1N1 | 2009/04/25–2009/06/20 | Week 8 | 463 | 122 | 127 | 189 | 262 | 218 |
| COVID-19 | 2020/03/11–2020/05/06 | Week 8 | 1,989 | 656 | 697 | 1,090 | 1,102 | 138 |
| Proportion | 4.30 | 5.38 | 5.49 | 5.77 | 4.21 | 0.63 | ||
| H1N1 | 2009/04/25–2009/06/27 | Week 9 | 534 | 141 | 147 | 216 | 307 | 245 |
| COVID-19 | 2020/03/11–2020/05/13 | Week 9 | 2,502 | 830 | 879 | 1,355 | 1,388 | 161 |
| Proportion | 4.69 | 5.89 | 5.98 | 6.27 | 4.52 | 0.66 | ||
| H1N1 | 2009/04/25–2009/07/04 | Week 10 | 582 | 158 | 165 | 240 | 330 | 262 |
| COVID-19 | 2020/03/11–2020/05/20 | Week 10 | 3,101 | 1,024 | 1,093 | 1,670 | 1,731 | 203 |
| Proportion | 5.33 | 6.48 | 6.62 | 6.96 | 5.25 | 0.77 |
Each theme was individually analyzed and no exclusion was done using a specific theme as dominant. The data search was carried out using the PubMed (Public/Publisher MEDLINE) (https://pubmed.ncbi.nlm.nih.gov - Date - completion) for: (i) (swine flu OR H1N1 OR H1N1 influenza virus OR influenza virus OR H1N1 influenza OR H1N1/09 OR H1N1 virus OR A(H1N1)pdm09 OR H1N1 flu OR Mexican flu OR influenza A); (ii) (coronavirus disease-19 OR coronavirus disease OR corona virus OR COVID-19 OR COVID19 OR SARS-CoV-2); (iii) the descriptors of (i) and (ii) were used along with the following descriptors: (iiia) AND (diagnosis); (iiib) AND (symptoms); (iiic) AND (epidemiology); (iiid) AND (treatment); (iiie) AND (vaccine). Data extraction was carried out weekly for ten weeks after the WHO declared the swine flu (25th April 2009) and COVID-19 (11th March 2020) diseases as pandemic. Only published studies written in English (filter 1) and about Human species (filter 2) were evaluated.
The proportion between the number of published articles for COVID-19 and H1N1 pandemics was also set in our data. Only published studies written in English (filter 1) and about Human species (filter 2) were evaluated. In addition, in the PubMed, the advanced search tool was used to limit the time to collect the number of studies as previously described and the terms “Date - completion” were considered to give only the information about the studies published as their final version. In brief, the number of articles published for both pandemics and the proportion between them are shown in Table 1.
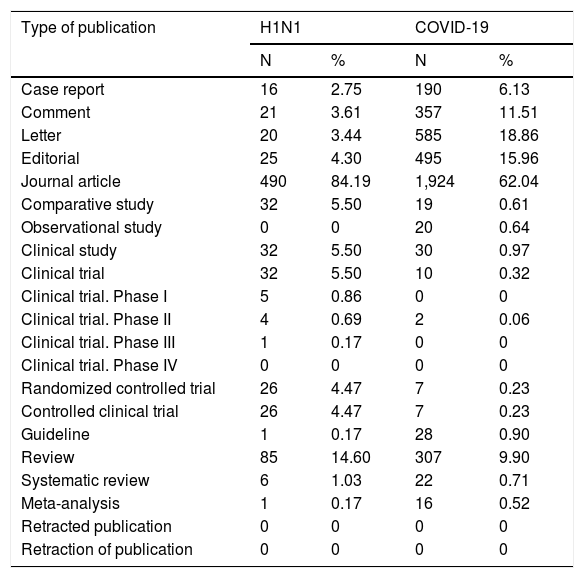
To improve the information about the studies published during both pandemic periods, the number of articles collected throughout the ten weeks after the WHO declared the swine flu and COVID-19 diseases as pandemic were categorized using the description for article type from PubMed as follows: case report, comment, letter to the editor, editorial, journal article, comparative study, observational study, clinical study, clinical trial (phase I, phase II, phase III or phase IV), randomized controlled trial, controlled clinical trial, guidelines, review, systematic review, meta-analysis, retracted publication or retraction of publication (Table 2). The percentage of each type of study was calculated based on the total number of articles published during each follow-up period.
Types of publications for the studies about swine flu disease (2009) and COVID-19 (2020) disease ten weeks after the World Health Organization declared both diseases as pandemic.
| Type of publication | H1N1 | COVID-19 | ||
|---|---|---|---|---|
| N | % | N | % | |
| Case report | 16 | 2.75 | 190 | 6.13 |
| Comment | 21 | 3.61 | 357 | 11.51 |
| Letter | 20 | 3.44 | 585 | 18.86 |
| Editorial | 25 | 4.30 | 495 | 15.96 |
| Journal article | 490 | 84.19 | 1,924 | 62.04 |
| Comparative study | 32 | 5.50 | 19 | 0.61 |
| Observational study | 0 | 0 | 20 | 0.64 |
| Clinical study | 32 | 5.50 | 30 | 0.97 |
| Clinical trial | 32 | 5.50 | 10 | 0.32 |
| Clinical trial. Phase I | 5 | 0.86 | 0 | 0 |
| Clinical trial. Phase II | 4 | 0.69 | 2 | 0.06 |
| Clinical trial. Phase III | 1 | 0.17 | 0 | 0 |
| Clinical trial. Phase IV | 0 | 0 | 0 | 0 |
| Randomized controlled trial | 26 | 4.47 | 7 | 0.23 |
| Controlled clinical trial | 26 | 4.47 | 7 | 0.23 |
| Guideline | 1 | 0.17 | 28 | 0.90 |
| Review | 85 | 14.60 | 307 | 9.90 |
| Systematic review | 6 | 1.03 | 22 | 0.71 |
| Meta-analysis | 1 | 0.17 | 16 | 0.52 |
| Retracted publication | 0 | 0 | 0 | 0 |
| Retraction of publication | 0 | 0 | 0 | 0 |
N, number of studies; %, percentage related to the total number of studies published after ten weeks of follow-up period. H1N1, Influenza A virus subtype H1N1; COVID-19; Coronavirus Disease 2019 (COVID-19). The types of studies were obtained from the PubMed database according to their classification.
The number of published articles about each virus or pandemic showed different numbers of publications during the first weeks after the WHO declared the two diseases a pandemic. The difference for the number of articles between pandemics improved because the number of studies about COVID-19 presented a faster increase, achieving four-times the number of publications about the swine flu pandemic from week 7 to week 10 after the WHO declared the swine flu and COVID-19 as pandemic. The same result was found when the studies were grouped for diagnosis, symptoms, epidemiology and treatment. However, the “vaccine” term was associated with a lower number of publications for COVID-19 pandemic when compared with the swine flu pandemic, showing only ∼0.48 of the number of studies during the first two weeks after the WHO declared the diseases as pandemics. However, the number of publications for COVID-19 pandemic achieved the mark of ∼0.77 when compared to swine flu pandemic during the week 10.
Science is crucial to promoting knowledge based on evidence and appears as a central pillar during critical events such as the two pandemics described in our data. The time lapse between the two events (pandemics) was only ten years and differences occurred in the number of publications considering the topics concerned. The higher number of studies published during the second evaluated period, namely COVID-19 pandemic, is evident. However, also noticeable is the higher number of case reports (N = 190), comments (N = 357), editorials (N = 495) and letters to the editors (N = 585) during the COVID-19 pandemic which increased the number of studies during this pandemic. These types of papers represent the lowest levels of the evidence pyramid showing a higher risk of bias,5 also, these types of studies promote the possibility of fast discussion and dialogue among specialists, favoring better insights for future investigations. Moreover, the number of studies including the need for individual protection equipment and social isolation to control the dissemination of the SARS-CoV-2 virus is evident in the literature, highlighting the need of psychological care.5–8 Likewise, the number of reviews, systematic reviews and meta-analysis including information about the risk factors related with the severity of the SARS-CoV-2 virus infection was higher than for H1N1 infection mainly regarding epidemiological data and/or comorbidities as risk factor for severe affection.9–12 In addition, the number of clinical trials was greater for H1N1 pandemic, and the clinical trial (phase 2) during the first ten weeks of COVID-19 presented information about the use of Chloroquine Diphosphate as Adjunctive Therapy.13 Also, in the first ten weeks after both pandemics were declared, no study was retracted. However, the Lancet journal retracted the study entitled “Hydroxychloroquine or Chloroquine With or Without a Macrolide for Treatment of COVID-19: A Multinational Registry Analysis” by Mehra et al.14 and the Lancet editors expressed some concern about that study’s data validity.15
The SARS-CoV-2 virus identification for COVID-19 was evidentiated in studies showing its limitations and highlights. However, it is necessary to optimize it to improve the applicability of this knowledge in several countries, such as Brazil, where the diagnostic test for SARS-CoV-2 is only carried out for severe cases of COVID-19.4,16 Moreover, in the treatment area, the COVID-19 pandemic was associated with the hydroxychloroquine or chloroquine dilemma including divergencies between governments and the WHO; since hydroxychloroquine or chloroquine still has not been considered an efficient therapy for COVID-19 disease and the WHO discontinued the studies for these drug as COVID-19 therapy.4,17–19 The epidemiology and symptoms were widely explored for COVID-19 pandemic and gave us the opportunity to understand the disease affection including cases with rare symptoms; moreover, the scientific community was able to determine the disease dissemination in a globalized world which facilitated the contamination between inhabitants from different countries and/or continents.18,19 The vaccine until now is not available for SARS-CoV-2 virus infection, however, studies with good results were published and we hope to have a supply of vaccines as soon as possible.18,20 Despite all limitations for better treatment for COVID-19, some discoveries highlight the importance of science, and among the important discoveries, it seems relevant to emphasize that in only four months the first randomized clinical trial with remdezivir was designed, conducted and published, proving that science can proceed very fast when under pressure.21 Currently, there is a need for the valorization of science and the practical use of findings based on studies with greatest level of evidence and lowest risk of bias.
Brazilian scientists suffer from low financial support for science and the COVID-19 pandemic evidenced the low importance given by the government to this area. The Brazilian President favors popular beliefs to treat the disease, minimizing the risk of infection and the severity associated with this illness; however, this week the Brazilian President was diagnosed with SARS-CoV-2 infection. Brazilian scientists and health professionals wish the president a quick recovery and we trust that the scientific work will be better recognized regarding the efforts made to control the pandemic and to treat these patients.
In conclusion, science give us the knowledge to deal with situations such as the pandemics. However, in some countries, including Brazil, science should be better valued by the governments and the community. Special attention should be paid to the number of published studies hurriedly with low level of evidence and high risk of bias.22 Readers should always evaluate the quality of each study and have a critical point of view to put into practice the knowledge acquired from the publications.
Conflicts of interestThe authors have no conflicts of interest to declare.