The therapeutic value of early physiotherapeutic treatment in critical respiratory settings has already been clearly outlined in the last fifteen years by several authors. However, there is still a controversial perception of mobilisation by healthcare professions.
In-bed cycling has attracted increasing attention having been demonstrated as a feasible and safe intervention in critical settings. Patients with respiratory diseases are typically prone to fatigue and exertional dyspnoea, as we observe in COVID-19 pandemic; in fact, these patients manifest respiratory and motor damage that can even be associated with cognitive and mental limitations. COVID-19 is at risk of becoming a chronic disease if the clinical sequelae such as pulmonary fibrosis are confirmed as permanent outcomes by further analysis, particularly in those cases with overlapping pre-existent pulmonary alterations.
In the present article, we propose a practical analysis of the effects of in-bed cycling, and further discuss its potential advantages if used in critical patients with COVID-19 in intensive care settings.
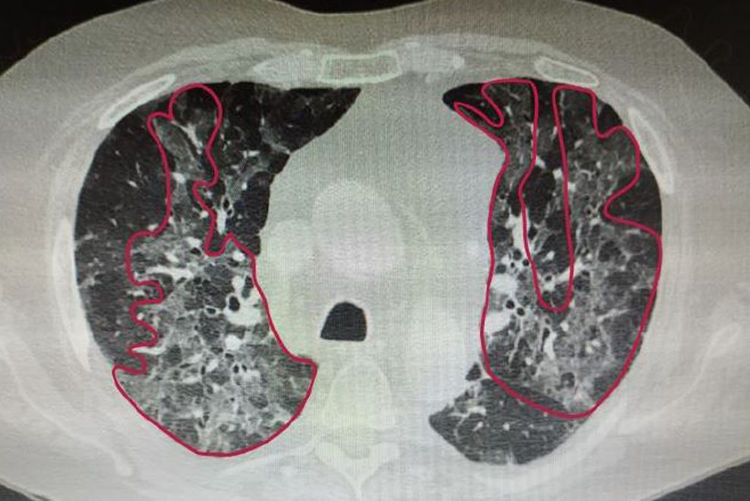
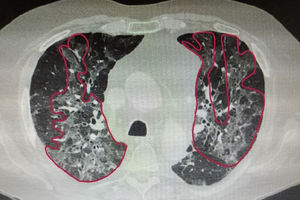
A 75-years-old man with a body mass index of 26.1...kg/m2 came to our attention having several comorbidities (hypertension, dyslipidemia, bronchial asthma, ulcerative colitis, critical stenosis of both the right coronary artery and the anterior intraventricular artery treated with angioplasty and stenting) and a 40 pack/year smoking history. Before hospitalisation, his daily autonomy was limited by the reduced walking capacity resulting from an accidental fall at home requiring bed rest because of limb pain and soreness. The patient went to the emergency room given the persistence of dyspnoea, cough and fever, although therapy with antibiotics and steroids had been administered at home. Hemogasanalysis showed moderate gas exchange impairment (PaO2/FiO2...=...238 mmHg and A-a O2 gradient...=...50...mmHg). The chest high-resolution computed tomography demonstrated the presence of multiple large areas of peri-bronchial ground glass compatible with bilateral interstitial pneumonia (Fig. 1). Blood tests demonstrated elevation of phlogosis indices and associated lymphopenia: Protein-C-Reactive (8.2...mg/dL), IL-6 (160.9...pg/mL), High Ferritinemia (1098...ng/mL), Lactate dehydrogenase (461 U/L), Negative Procalcitonin (0.1...ng/mL). Given the suggestive picture, a nasal/oropharyngeal swab for SARS-Cov-2 was performed and resulted positive. This case describes a patient with severe respiratory failure in bilateral interstitial pneumonia associated with critical deterioration of the physical function because of prolonged bed rest.
The patient was initially admitted to a general ward; gas-exchanges gradually deteriorated (PaO2/FiO2...=...115...mmHg). He needed oxygen therapy with Ventimask and reservoir to maintain sufficient oxygenation. Given the severe deterioration, high-dose steroid therapy was also initiated. On hospital day 8, the patient was transferred to the Respiratory Intensive Care Unit (St Orsola University Hospital, Bologna, Italy) starting high flow oxygen therapy (flow 50...L/min ... FiO2 43%) because of the persistence of severe gas exchange impairment. Given the respiratory failure, medical treatment was completed with Ruxolitinib, a selective Janus Associated Kinases inhibitor usually used for haematological diseases1,2. There was a gradual improvement in gas-exchanges (PaO2/FiO2...=...152, 236, 278...mmHg on hospital days 18, 20, and 21, respectively), allowing suspension of oxygen therapy. Severe sarcopenia contributed to disability and reduced ability to cope with the stress of an illness, in the present case3.
Physiotherapeutic treatmentPhysical evaluation revealed difficulties in getting into the sitting position on the edge of the bed autonomously; assistance was necessary to complete the postural passage from a supine to a sitting position. Marked hypotrophy of lower extremities was also present; hip flexion, knee extension, and ankle dorsiflexion were 3/5 as assessed by the Medical Research Council Muscle Scale4. A trial for a passage from a sitting to a standing position revealed the patient could not finalise the movement even with assistance of two operators. The patient started a physiotherapeutic programme consisting of in-bed motor exercises and specific postural transfer training, aiming to achieve sitting position as soon as possible.
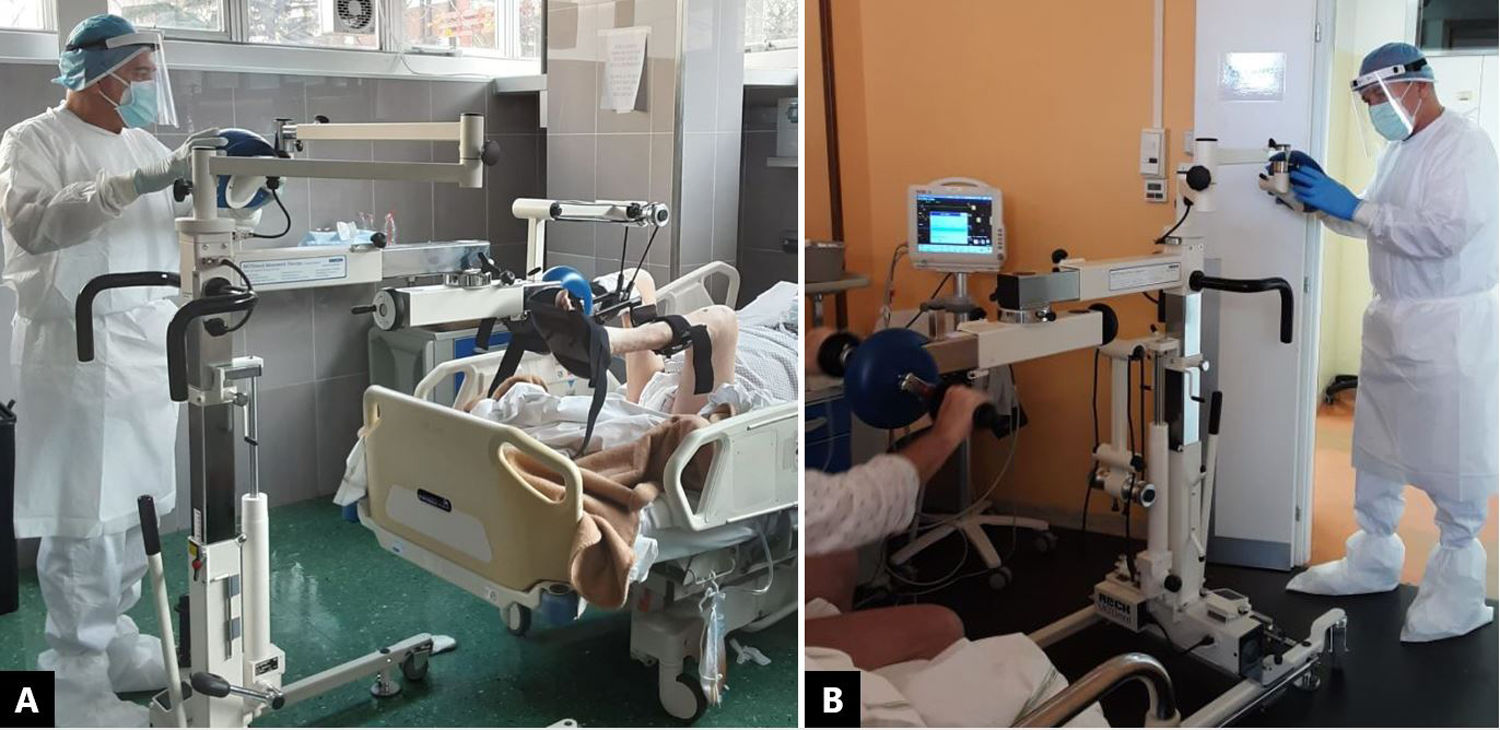
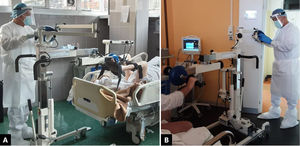
Considering the clinical frame and the concomitant positivity to SARS-CoV-2, it was decided to use in-bed electrical cycle ergometry (MOTOmed letto2, RECK-Technik GmbH & Co. KG, Betzenweiler, Germany) to facilitate and enhance lower limb function. The device is designed to provide training for both upper and lower extremities in a passive, motor-assisted, and active mode directly from the hospital bed (Fig. 2). The patient had one daily session lasting ...30...min, depending on clinical conditions. The exercise was supervised and was to be interrupted in the case of increased heart rate (>20 bpm from the baseline) or subjective fatigue/dyspnoea higher than moderate ...scoring 3... in the modified Borg Scale5. Active movements of upper and lower limbs were also proposed, and the patient was asked to repeat exercises autonomously during the day while in a sitting position. The patient kept to all therapeutic sessions, and no adverse events related to treatment were observed. The patient was discharged to a low-intensity setting of care on hospital day 26, being able to maintain a sitting position and having increased lower limb mobility although not yet able to reach a standing position.
Rehabilitation in critical settings: from the origins to 2020The positive effects of early mobilisation were first described in the last century when Dr Powers, in his report, illustrated that benefits occurred in a series of 100 patients undergoing different types of surgery6. In that study, the treatment consisted of encouraging patients to sit out of bed and to walk already on the first postoperative day, assuming such activities would bring significant advantages in enhanced recovery and restoration of daily autonomy. The study results did indeed demonstrate that all those patients treated with early mobilisation had reduced number of days in bed, length of stay and convalescence duration in weeks.
Enhanced recovery started to be discussed as early as in the 1940s6; nevertheless, there were no defined and specific programmes available for implementation in critical respiratory settings until the first studies were published in the mid-1990s7,8. If early postoperative rehabilitation is a valuable intervention, from that study it can be gathered that a structured rehabilitation programme is feasible, safe, and effective to counteract the physical impairment and to restore functional ability in those patients who are prone to deconditioning related to respiratory failure8. It should be highlighted that the main differences between postoperative rehabilitation and pulmonary rehabilitation are primarily associated with the presence of the underlying respiratory disease, which poses several challenges in terms of medical and rehabilitative issues and hospital readmission rates9. At the same time, it should not be forgotten that pulmonary rehabilitation is a multidisciplinary intervention involving several medical professionals to manage all the clinical aspects connected to respiratory-related physical and psychological limitations10,11.
The therapeutic value of early physiotherapeutic treatment in critical respiratory settings has already been clearly outlined in the last fifteen years by several authors12...18; despite this, there is still a controversial perception about mobilisation by healthcare professions19.
Searching literatureWe used in-bed cycling; to date, no studies have researched the physiological effects of this type of assisted mobilisation in patients with COVID-19. However, in-bed cycling has attracted increasing attention, having been demonstrated as a feasible and safe intervention in critical settings20. To retrieve evidence regarding the use of in-bed cycling we searched PubMed and Scopus databases using the following key words ..úin-bed cycling..., ..úcritical patients..., and ..úmotomed letto2.... Databases were searched from their inception until December 2020; we considered eligible only those citations written in English, Spanish, Italian or French. No limits were imposed for age, gender, and article types. We also searched the references of retrieved papers, and we made a further analysis of electronic search engines (i.e., Google) retrieving additional information.
Effects of in-bed cyclingPatients with respiratory diseases are typically prone to fatigue and exertional dyspnoea as we observe in COVID-19; in fact, these patients manifest respiratory and motor damages that can even be associated with cognitive and mental limitations21. COVID-19 is at risk of becoming a chronic disease if the clinical sequelae such as pulmonary fibrosis, are confirmed as permanent outcomes by further analysis22...27 particularly in those cases overlapping pre-existent pulmonary alterations28. Patients forced to prolonged bed rest or presenting with mobility restrictions, may simply develop muscle dysfunction or a more severe condition, namely intensive care unit acquired weakness, characterised by generalised muscle weakness affecting peripheral and respiratory muscles29,30. Loss of muscle mass is negatively correlated with 2...3 weeks of immobilisation or intensive care unit stay31. That said, in all those conditions exposing patients to prolonged or forced restricted mobility or ...even worse ... immobility, primary rehabilitative goals are preserving, and hopefully, restoring a certain degree of functional capacity. COVID-19 has posed several barriers to implementing traditional physiotherapeutic activities because of the need to contain infection risk among healthcare professionals. Particularly at the beginning of the pandemic, with reduced availability of personal protective equipment, proximity with patients was conditioned by considering the risks and benefits of specific procedures, including mobilisation. A specific research line investigating characteristics and indications of personal protective equipment has contributed to improving personal protection among healthcare workers over time32...34.
Can in-bed cycling in patients with COVID-19 be compared to treatments traditionally adopted in intensive care settings?
In a study by Ringdal et al., conducted to investigate physiological effects on in-bed cycling, participants ... who were patients hospitalised in an intensive setting ... did not show significant and/or alarming changes in physiological parameters while exercising. Heart rate, mean arterial pressure, respiratory rate, and peripheral oxygen saturation were confined to a small range of variation, confirming in-bed cycling to be safe and feasible35. In the same way, participants did not manifest cardiac rhythm changes such as atrial fibrillation or ventricular tachycardia; in that study, the in-bed cycle was a suitable means for enhancing recovery and promoting motor activities in those patients who were not able to perform postural transfers35. Additional physiological effects were related to interrupting sedation while cycling, allowing patients to be aware of their physical activity. Other aspects of in-bed cycling executed in critical settings are related to the patient...s perception of a feeling of control, safety, and hope of recovery during a critical illness35.
Several studies have shown that in-bed cycling enhances physical function, helping to prevent muscle atrophy, and it is a feasible intervention to get patients accustomed to exercise and mobility (Table 1).
Effects of in-bed cycling.
| Authors, ref. and year | N. of patients, [setting] | Main outcomes |
|---|---|---|
| Woo et al.39 2018 | 10, [ICU] | Increase of the rectus femoris muscle cross-sectional area, and augmented circumference of the thigh |
| Fossat et al.38 2018 | 158, [medical-surgical ICU] | The authors did not find significant differences regarding muscle strength between those patients who received in-bed cycling and electrical nerve stimulation and controls, as assessed by the MRC scale. Nor were differences between groups found for the following variables: 1) functional autonomy at discharge as assessed by the ICU Mobility Scale and the Katz Index of Independence in ADL, 2) number of ventilator-free days, 3) thickness of the rectus femoris |
| Machado et al.40 2017 | 22, [general-cardiac ICU] | Increase of the peripheral muscle strength as assessed by the MRC scale |
| Rocca et al.41 2016 | 9, [neurological ICU] | Increase in catecholamine production in patients treated with in-bed cycling |
| Burtin et al.36 2009 | 31, [medical-surgical ICU] | At ICU discharge the following variables were higher in patients who underwent in-bed cycling: the quadriceps force (N...kg), the walked distance (6MWT), the SF-36 PF score |
Legend: ICU, intensive care unit; MRC, Medical Research Council; ADL, activities of daily living; N...kg, Newton...kilogram; 6MWT, 6-min walking test; SF-36 PF, Short Form-36 physical functioning.
In addition, in-bed cycling has been found to improve consciousness and perception of personal mobility potential, emphasising another important aspect related to the hospital stay in critical settings where patients are often subjected to sedation and mechanical ventilation21,41.
Technical aspects of assisted mobilisation with in-bed cycle ergometersIn-bed cycling with the ergometer MOTOmed letto2 (RECK-Technik GmbH & Co. KG, Betzenweiler, Germany) ... the most widely described in the literature ... 21,36...43 can be adopted for exercising both upper and lower extremities either in a supine or in a sitting position (Fig. 2). The cycle ergometer device can be placed either laterally or at the end of the bed and firmly anchored to the floor thanks to a blocking mechanism ... preventing unwanted movements of the machine during the exercise session. The hands and legs grips/pedals are adjustable and facilitate patient positioning even in those subjects with reduced autonomous movement. The lower limbs are sustained by supports, and the feet are secured by velcro strips; hand pockets are also available for those unable to grip by themselves. The possibility of choosing a preferred exercise position allows this system to be used even in reduced spaces. The machine is equipped with wheels and can be easily moved within the ward. The cycle ergometer detects the patient...s active contribution and adjusts passive or active cycling accordingly. A digital controller mounted on a movable arm allows maintaining a distance from the patient ... while exercising ... of approximately two meters. This feature is handy when working in COVID-19 settings because it guarantees a safe distance for the operator (Fig. 2). Once the patient has been positioned for executing the preferred exercise, they can be supervised without the need to be in strict contact; meanwhile, the physiotherapist can treat other patients nearby. When the use of in-bed cycling is optimised to the workload, one can have more slots available. Depending on the patient...s clinical condition, when the cycle ergometer is placed laterally, the subject is facilitated to exercise the upper extremities and control the trunk position. Such modality is expected to enhance mobility and proprioceptive perception of the body posture requiring a certain degree of collaboration by the patient. In a way, when exercise is proposed in a sitting position on the edge of the bed, there are several advantages such as 1) improved active participation of the subject, 2) increased work of the antigravity muscles (i.e., paravertebral and abdominal groups) which can be particularly useful during weaning from bed rest related to other respiratory conditions such as either invasive and noninvasive mechanical ventilation, 3) augmented aerobic effort because of the possibility of increasing the speed, duration, intensity and type of exercise ... passing from a passive/assisted to an active modality during the same session.
RT300-SUPINE (https://www.cyclonemobility.com/product/rt300-supine-2; Cyclone Mobility, Widnes, UK) is another cycle ergometer that incorporates functional electrical stimulation using adhesive pads. The device is mounted on an expandable wheeled table and provides arm and leg cycling; adjustable housings are present for optimising arm and leg exercise, while a dedicated system controls the legs... lateral movements during cycling. The use of the RT300-SUPINE has been described in the study by Kho et al.44.
The THERA-Trainer Bemo (https://thera-trainer.com/en/thera-trainer-products/cycling/thera-trainer-bemo; medica Medizintechnik GmbH, Hochdorf, Germany) is another device for passive, assisted and active movements of upper and lower extremities. It is a wheeled structure that allows patients to exercise either in a supine or in a sitting position, and also provides visual feedback during the session. The device can be adjusted using the control and display unit; it includes emergency-stop functions and a system detecting spasticity.
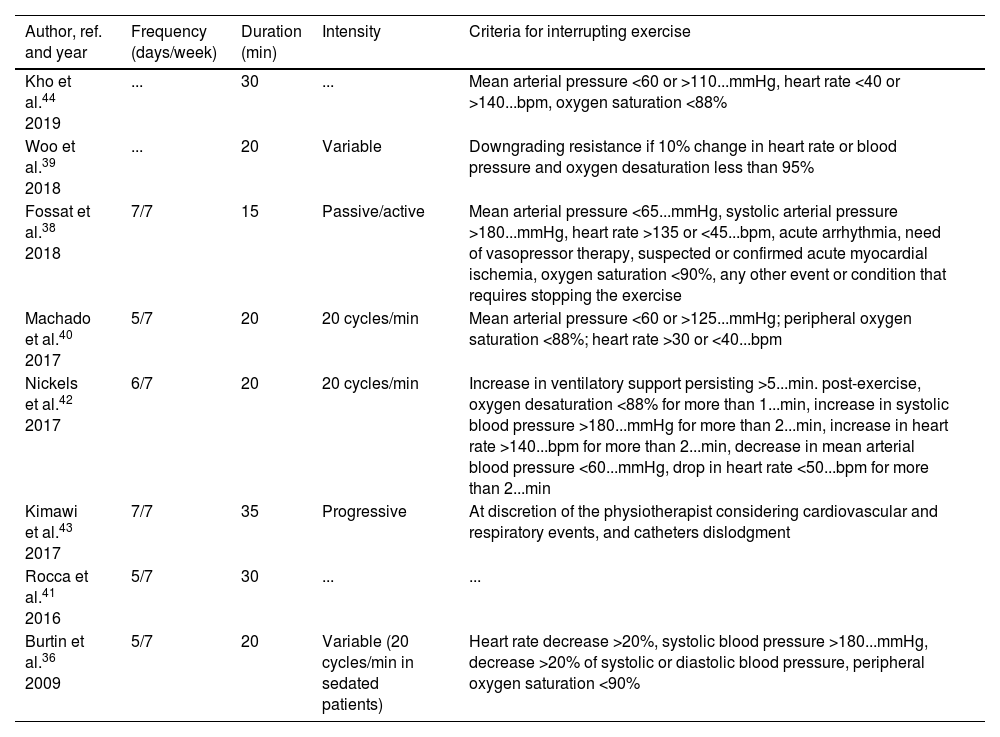
Treatment...s details of in-bed cyclingConsidering the evidence available, we gathered some common treatment schemes regarding duration, type, time, and intensity of exercise using in-bed cycling, as shown in Table 2. From these studies, it was possible to decide which criteria should be used for interrupting training; standard hemodynamic parameters such as heart rate, arterial pressure, and oxygen saturation should be used to guarantee safety during in-bed cycling.
In-bed cycling: training protocols.
| Author, ref. and year | Frequency (days/week) | Duration (min) | Intensity | Criteria for interrupting exercise |
|---|---|---|---|---|
| Kho et al.44 2019 | ... | 30 | ... | Mean arterial pressure <60 or >110...mmHg, heart rate <40 or >140...bpm, oxygen saturation <88% |
| Woo et al.39 2018 | ... | 20 | Variable | Downgrading resistance if 10% change in heart rate or blood pressure and oxygen desaturation less than 95% |
| Fossat et al.38 2018 | 7/7 | 15 | Passive/active | Mean arterial pressure <65...mmHg, systolic arterial pressure >180...mmHg, heart rate >135 or <45...bpm, acute arrhythmia, need of vasopressor therapy, suspected or confirmed acute myocardial ischemia, oxygen saturation <90%, any other event or condition that requires stopping the exercise |
| Machado et al.40 2017 | 5/7 | 20 | 20 cycles/min | Mean arterial pressure <60 or >125...mmHg; peripheral oxygen saturation <88%; heart rate >30 or <40...bpm |
| Nickels et al.42 2017 | 6/7 | 20 | 20 cycles/min | Increase in ventilatory support persisting >5...min. post-exercise, oxygen desaturation <88% for more than 1...min, increase in systolic blood pressure >180...mmHg for more than 2...min, increase in heart rate >140...bpm for more than 2...min, decrease in mean arterial blood pressure <60...mmHg, drop in heart rate <50...bpm for more than 2...min |
| Kimawi et al.43 2017 | 7/7 | 35 | Progressive | At discretion of the physiotherapist considering cardiovascular and respiratory events, and catheters dislodgment |
| Rocca et al.41 2016 | 5/7 | 30 | ... | ... |
| Burtin et al.36 2009 | 5/7 | 20 | Variable (20 cycles/min in sedated patients) | Heart rate decrease >20%, systolic blood pressure >180...mmHg, decrease >20% of systolic or diastolic blood pressure, peripheral oxygen saturation <90% |
In one study by Machado et al., the authors even provided the physiotherapist-to-patient ratio 1:8 returning an interesting consideration on physiotherapists... workload40.
Synthesising data from Table 2, exercise frequency can range between 5 to 7 days a week, duration between 5 to 35...min, with variable intensity ... passive, assisted, active ... depending on patients... clinical conditions.
Is in-bed cycling feasible and safe?Exercise with an in-bed cycle ergometer has been demonstrated to be feasible and safe, as outlined in several types of research20,37,43,44. In a recent study, overall comments from patients, clinicians, and family members were positive when recalling cycling sessions; all were concordant, affirming that in-bed cycling contributed to physical recovery20.
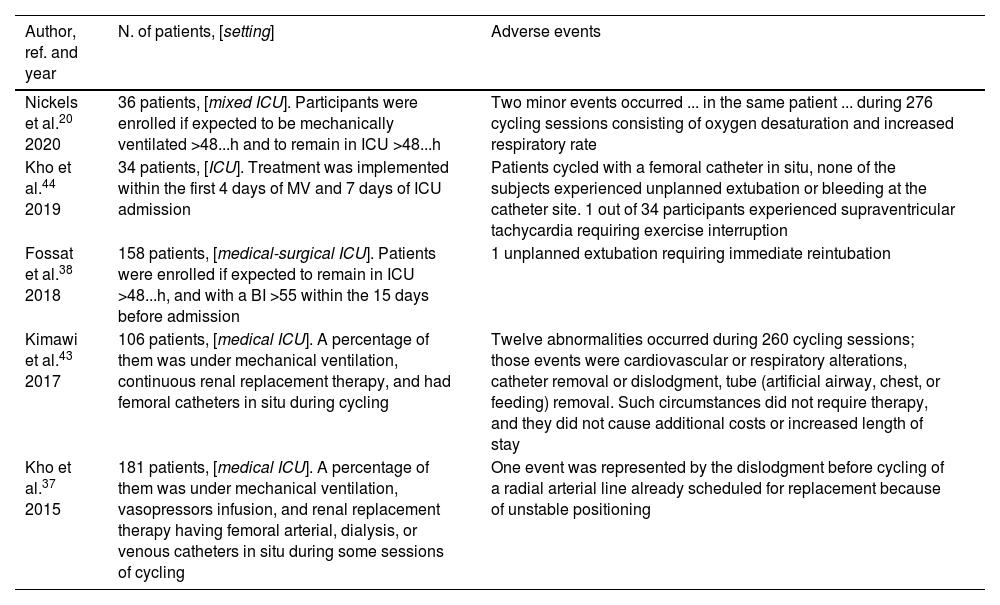
Among the effects of in-bed cycling, the sympathetic system stimulation has been found by Rocca et al. in patients with severe neurological injury41; such a stimulation seems to be recognised as a mechanism leading to increased production of catecholamines. The authors concluded that in-bed cycling, in patients with subarachnoid haemorrhage, should be used with caution because of the potential risk of vasospasm due to elevated catecholamine levels. However, they did not find contraindications to its use to prevent polyneuromyopathy in critical illness41. In intensive care settings, in-bed cycling should be supervised to guarantee safety during exercise at all times (Table 3).
In-bed cycling: adverse events reported in the literature.
| Author, ref. and year | N. of patients, [setting] | Adverse events |
|---|---|---|
| Nickels et al.20 2020 | 36 patients, [mixed ICU]. Participants were enrolled if expected to be mechanically ventilated >48...h and to remain in ICU >48...h | Two minor events occurred ... in the same patient ... during 276 cycling sessions consisting of oxygen desaturation and increased respiratory rate |
| Kho et al.44 2019 | 34 patients, [ICU]. Treatment was implemented within the first 4 days of MV and 7 days of ICU admission | Patients cycled with a femoral catheter in situ, none of the subjects experienced unplanned extubation or bleeding at the catheter site. 1 out of 34 participants experienced supraventricular tachycardia requiring exercise interruption |
| Fossat et al.38 2018 | 158 patients, [medical-surgical ICU]. Patients were enrolled if expected to remain in ICU >48...h, and with a BI >55 within the 15 days before admission | 1 unplanned extubation requiring immediate reintubation |
| Kimawi et al.43 2017 | 106 patients, [medical ICU]. A percentage of them was under mechanical ventilation, continuous renal replacement therapy, and had femoral catheters in situ during cycling | Twelve abnormalities occurred during 260 cycling sessions; those events were cardiovascular or respiratory alterations, catheter removal or dislodgment, tube (artificial airway, chest, or feeding) removal. Such circumstances did not require therapy, and they did not cause additional costs or increased length of stay |
| Kho et al.37 2015 | 181 patients, [medical ICU]. A percentage of them was under mechanical ventilation, vasopressors infusion, and renal replacement therapy having femoral arterial, dialysis, or venous catheters in situ during some sessions of cycling | One event was represented by the dislodgment before cycling of a radial arterial line already scheduled for replacement because of unstable positioning |
Legend: ICU, intensive care unit; MV, mechanical ventilation; BI, Barthel Index.
In-bed cycle ergometry seems to be a promising intervention in different types of patients including intensive care settings, as outlined in several research studies. Although such a trend has been confirmed in published studies, up until now no clinical experiments have described the use of in-bed cycling in patients with COVID-19. Considering the knowledge and evidence gathered from literature, we feel there is enough to corroborate the hypothesis that in-bed cycling can be used in COVID settings with selected patients (i.e., respiratory-related motor impairment, sarcopenia, and muscle hypotrophy related to prolonged bed rest). In addition to the therapeutic benefits for patients, in-bed cycling allows healthcare professionals to distance while operating, and this is not of secondary importance in such a pandemic. Additional advantages can be found in delegating exercise supervision to different multidisciplinary team members ... once instructed ... freeing specific staff ... such as physiotherapists ... for other simultaneous interventions, particularly when human resources are lacking or scarce.
FundingNone.
Conflict of interestsThe authors have no conflicts of interest to declare.
We would like to thank Cecilia Mari for revising the use of English in the present manuscript.
Series: How I manage the case. Series Editor: Stefano Nava.