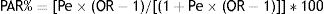Asthma is a chronic and heterogeneous disease that affects people of all ages and has a high estimated increase in prevalence worldwide. Asthma control represents a main goal in the disease management. International studies revealed low levels of disease control resulting in a significant burden for healthcare systems, not only in terms of quality of life, but also in terms of health costs. Modifiable and non-modifiable factors have been identified as relating to poor asthma control level. In this study we evaluated the distribution of asthma control levels in Portuguese adult population and examine the determinants associated with uncontrolled asthma.
Materials and methodsUsing a similar methodology to the one employed in the Asthma Insights and Reality in Europe (AIRE) survey, 327 active asthmatic patients were identified by random phone number and completed a questionnaire during 2011 to 2012. Asthma control was assessed by the evaluation of GINA based symptom control, by Asthma Control Test.äó (ACT) and by self-perception of control. To examine the relationship between uncontrolled asthma and its determinants, univariate logistic regression analysis, sequential multivariable regression and population attributable risk percentage were determinate.
Results35.2% active asthmatic patients had uncontrolled asthma, 64.8% partially controlled and none of the individuals had total control of asthma assessed by ACT test. Factors significantly associated with poor asthma control scores were: age (OR 1.02 per year of age; 95% CI: 1.01.Çô1.03), female sex (OR 1.87; 95% CI: 1.15.Çô3.04), educational level (OR 0.5; 95% CI: 0.28.Çô0.89 at high school level or over), occupation (OR 4.92; 95% CI: 2.12.Çô11.42 if looking for a first job or unemployed) (OR 2.51; 95% CI: 1.35.Çô4.65 if being retired), income (OR 0.23; 95% CI: 0.07.Çô0.72 if >619 euros), BMI (OR 1.09 per BMI unit; 95% CI: 1.03.Çô1.14), having rhinitis symptoms (OR 4.40; 95% CI: 2.56.Çô7.58) and using inhaled corticosteroids (OR 0.44; 95%CI: 0.24.Çô0.82 if used in the past or never used).
Looking for a first job or being unemployed, BMI and having rhinitis symptoms remained significant after multivariate adjustments.
ConclusionsUncontrolled asthma was associated with several determinants. Their identification can contribute to improve asthma care both from clinical and from public health perspectives.
Asthma is a chronic and heterogeneous disease1 that affects people from all ages and ethnicities worldwide.2 There is an estimated a global prevalence of 315 million people with asthma,3 contributing with more than 23 million of Disability-Adjusted Life Years (DALYs).4 Asthma prevalence has been increasing in many countries, in parallel with the development of urbanization and the adoption of modern lifestyles.5 It is estimated that by 2025 100 million of people more will have asthma worldwide.5
Despite the existence of treatment guidelines, many individuals with asthma remain undertreated and uncontrolled, resulting in a significant burden, not only in terms of quality of life, but also in terms of health costs.2 Health cost associated with asthma could be direct costs, like treatments and hospitalizations, as well as indirect costs, due to work absences and premature death.2 In 2010, the yearly cost per asthmatic adult patient in Europe, according to estimates from Accordini et al.,6 ranged from 509.é¼ for a controlled asthma patient to 2281.é¼ for an uncontrolled asthma patient.
The Global Initiative for Asthma (GINA) was created in 1993 with the objective of disseminating accurate information about asthma and improving the quality of asthma patient care worldwide.1 At present, according to GINA guidelines, achieving and maintaining asthma control represents one of the main goals for the management of the disease and consists of the evaluation of the control of symptoms and future risk of unfavourable outcomes.1
Numerous factors such as age, gender,7 comorbidities7,8 psychological aspects9 and socioeconomic determinants10 have been studied and reported as associated with asthma control. Behavioural reasons such as smoking habits7 or therapeutic compliance have also been related to poor asthma control levels.11
A multiway and a complex causal relationship between health, education level, work and income has been reported. Having a chronic disease can be considered a risk factor for employment. In Portugal, 66,294 of potential productive life years were lost due to non-communicable diseases (cardiovascular disease, diabetes, cancer and respiratory diseases) in 2013, according Organization for Economic Co-operation and Development (OECD).12
The Portuguese National Asthma Survey reported a proportion of 43.1% uncontrolled disease in asthmatic Portuguese population.13 In a national population study non-modifiable factors such as age and gender were associated with uncontrolled asthma.14 Self-evaluation of uncontrolled asthma, participation in decisions related to treatment and the presence of comorbidities have also been considered as important contributions to asthma control.14
The purpose of this study was to investigate the distribution of asthma control level in Portuguese adult population, using different approaches of control level evaluation, and to examine the determinants associated with uncontrolled asthma.
MethodsStudy designA cross-sectional study at national level (Portugal) was carried out from March 2011 to March 2012, based on the methodology applied in the Asthma Insights and Reality in Europe (AIRE).15
A probabilistic sample of individuals diagnosed with .Ç£active asthma.Ç¥ was identified through telephone numbers and a structured telephone interview was used. In order to select exclusively individuals with active asthma, an adult in each household was asked the following question: .Ç£Has a physician ever diagnosed any family member in your home as having asthma?.Ç¥. Participants were included as having active asthma if they answered positively at least one of the following questions: .Ç£Do any of those individuals diagnosed with asthma take any medication for their asthma?.Ç¥ and .Ç£Has any of those individuals diagnosed with asthma had any asthma crises or asthma symptoms in the last year?.Ç¥. If more than one household member with asthma was qualified, a respondent was randomly selected for interview.
A total of 557 individuals were identified with active asthma and 327 complete interviews were acquired in adults (.ëÑ16 years old).
Control assessmentAsthma control was evaluated in three ways: GINA based symptom control, by Asthma Control Test.äó (ACT) and individual self-perception of control assessment.
The asthma symptom control, based on GINA, included four questions, relating to the previous four weeks, about the frequency of daytime symptoms, night waking, relief medication needs and activity limitations due to asthma.
ACT is a quantitative self-assessment tool for asthma control, which consists of 5 simple questions for the interviewee to consider on the impact of asthma on a daily basis over the previous four weeks; relating to shortness of breath, use of rescue medication, nocturnal asthma symptoms and self-evaluation of asthma control.16 According to Thomas et al.17 ACT scores equal to or below 19 are considered as identifying patients with poorly controlled asthma, as defined by GINA. ACT is also used for the evaluation of determinants associated with poor asthma control. ACT scores equal to or below 19 points correspond to .Ç£uncontrolled.Ç¥ asthma and scores of at least 20 points are classified as .Ç£controlled.Ç¥ (20.Çô24=.Ç£partially controlled.Ç¥; 25=.Ç£totally controlled.Ç¥).
From the main questionnaire of the study, several determinants for the analysis were selected according to their relevance and the consulted bibliography: age, sex, civil status, region of residence, educational level, occupation, household income, body mass index (BMI), smoking habits, rhinitis symptoms, allergies as a trigger for crises, having family doctor, assistant physician to solve asthma problems, seasonal flu vaccination, Peak Flow Metre use, conducting lung function tests, training to use the inhaler properly, written asthma action plan, forgetting to take medication, inhaled corticosteroid use and number of medications used for asthma. BMI was classified according to the World Health Organization (WHO) criteria for adults,18 being underweight if BMI <18.5kg/m2, pre-obesity if 25.0kg/m2.ëÑBMI<30.0kg/m2, and obesity if BMI .ëÑ30.0kg/m2.
Statistical analysisDescriptive statistics were described as absolute frequency and proportion for categorical variables and continuous variables were represented as mean (standard deviation) for normally distributed variables and as median (interquartile range) for non-normally distributed variables.
The age differences between sexes was evaluated by the Mann Whitney U Test.
For additional analyses two groups of active asthma respondents were considered: controlled (ACT score .ëÑ20) and uncontrolled (ACT score .ëñ19).
To examine the relationship between uncontrolled asthma and its determinants, an indicator of uncontrolled asthma was used as a dependent variable. The determinants were classified into the following domains, grouping conceptually related variables: demographic, socioeconomic, risk and clinical factors, clinical care and treatment. Univariate logistic regression analysis was done for all determinants. From a public health perspective and in order to measure the contribution of each determinant to the level of asthma control, the population attributable risk percentage (PAR%) was assessed, using the following formula:
(Pe=estimate of population exposure)
with OR as the inverse risk (1/OR) in the case of a protective factor (odds ratio lower than 1).
Sequential multivariable regression was performed. Variables with p<0.30 in the univariate analysis were used to build the adjusted models (Forward Likelihood Ratio method). Multivariable adjustments were performed in sequence considering additions to variables for the different domains, where variables from a given domain were adjusted to the multivariable model, as noted in the Table 4. Pseudo r-squared (Cox and Nagelkerke) were calculated from the models originated.
For all tests performed, the level of statistical significance was set at 0.05. All analyses were conducted using SPSS version 23 (IBM Corp., Armonk, N.Y., USA).
ResultsGeneral characteristicsA total of 327 full interviews were completed with adults who had active asthma. A detailed description of the demographic and asthma related variables is summarized in Table 1. Further socioeconomic, risk and clinical factors, as well as clinical care and treatment characteristics are described below.
Respondent characteristics.
| Age, median (IQR) | 44 (27.Çô58) |
| Female, n (%) | 203 (62.1) |
| Age at diagnosis of asthma, median (IQR) | 15 (5.Çô36) |
| Duration of asthma in years, median (IQR) | 20 (10.Çô30) |
| Smoking habits (n=315) | |
| Non-smoker, n (%) | 199 (63.2) |
| Current smoker, n (%) | 49 (15.6) |
| Former smokers, n (%) | 67 (21.3) |
| Body mass index (n=310) | |
| Underweight, n (%) | 10 (3.2) |
| Normal weight, n (%) | 138 (44.5) |
| Pre-obesity, n (%) | 115 (37.1) |
| Obesity, n (%) | 47 (15.2) |
IQR: interquartile range.
In the sampled population, 62.1% were female and 37.9% male. In female the median age was 46 (29.Çô58) and in male 37.5 (26.Çô56), without statistic significant differences. A high percentage of the individuals questioned was pre-obese or obese (52.3%) and 36.9% were ever smokers.
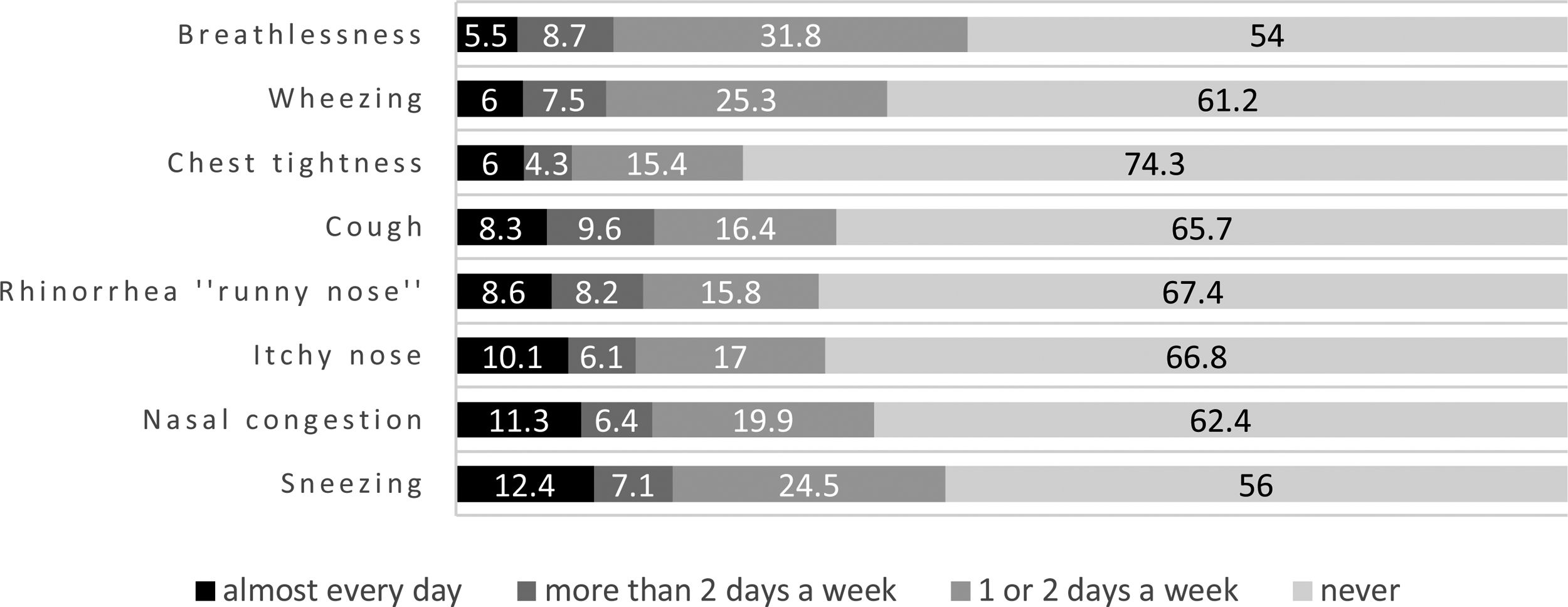
Asthma controlFrequency of self-reported symptoms (in the last four weeks) is discriminated in Fig. 1.
In relation to GINA based symptoms control assessment, of the 327 adult individuals interviewed, 18% had uncontrolled asthma, 47.7% partially controlled and 34.3% had totally controlled. Based on ACT test, 35.2% of the subjects had uncontrolled asthma, 64.8% partially controlled and none of the individuals had total control. 93.9% of the individuals perceived their asthma as completely, well or somewhat controlled and only 6.1% perceived their asthma poorly or not controlled at all (Table 2).
Asthma control assessment.
| GINA based symptom control | |
| Controlled, n (%) | 112 (34.3) |
| Partly controlled, n (%) | 156 (47.7) |
| Uncontrolled, n (%) | 59 (18.0) |
| ACT score | |
| Controlled asthma (ACT=25), n (%) | 0 |
| Partially controlled (ACT 20.Çô24), n (%) | 212 (64.8) |
| Non-controlled asthma (ACT .ëñ19), n (%) | 115 (35.2) |
| Patient perceived control | |
| Completely controlled, n (%) | 84 (25.7) |
| Well controlled, n (%) | 145 (44.3) |
| Somewhat controlled, n (%) | 78 (23.9) |
| Poorly controlled, n (%) | 18 (5.5) |
| Not controlled at all, n (%) | 2 (0.6) |
Concerning the agreement between ACT asthma control score and the participants.ÇÖ self-perception, about 39.2% of individuals with uncontrolled asthma evaluated by ACT self-reported their asthma as completely or well controlled and only 16.5% had the self-perception that agreed with the objective assessment of asthma control.
Determinants for uncontrolled asthmaWe then sought to understand which factors were associated with poor asthma control. In a univariate analysis, the factors significantly associated with poor asthma control scores (ACT score .ëñ19) were: age, female sex, educational level (Table 3). The PAR% for the significant variables are also in Table 3. In order to understand the relationship of significant variables in the univariate analysis a sequential multivariable adjustment analysis was performed by adding the dimensions of factors sequentially (Table 4). Occupation, BMI and having rhinitis symptoms were the factors that remained statistically significant results besides the sequential adjustments.
Univariate analysis for uncontrolled asthma and Population attributable risk %.
| Determinants | All participantsn (%) | Uncontrolled asthman (%) | Risk for uncontrolled asthma (ACT score .ëñ 19) | Population attributable risk % | ||
|---|---|---|---|---|---|---|
| Crude Odds Ratio (95% CI) | p Value | |||||
| Demographic | ||||||
| Age, classes | 16.Çô25 | 67 (20.5%) | 17 (14.8%) | ref | ||
| 26.Çô45 | 105 (32.1%) | 34 (29.6%) | 1.41 (0.71.Çô2.80) | 0.327 | .Çô | |
| 46.Çô65 | 109 (33.3%) | 42 (36.5%) | 1.84 (0.94.Çô3.61) | 0.074 | .Çô | |
| > 65 | 46 (14.1%) | 22 (19.1%) | 2.70 (1.21.Çô5.99) | 0.015 | 19.3% | |
| Age, years | 44 (27.Çô58)a | 47.69 (18.7)b | 1.02 (1.01.Çô1.03) | 0.005 | .Çô | |
| Sex | Male | 124 (37.9%) | 33 (28.7%) | ref | 0.012 | |
| Female | 203 (62.1%) | 82 (71.3%) | 1.87 (1.15.Çô3.04) | 35.1% | ||
| Civil status | Single | 106 (33.0%) | 34 (29.8%) | ref | ||
| Married or Cohabited | 174 (54.2%) | 62 (54.4%) | 1.17 (0.70.Çô1.96) | 0.543 | .Çô | |
| Divorced, separated or Widow (er) | 41 (12.8%) | 18 (15.8%) | 1.66 (0.79.Çô3.47) | 0.181 | .Çô | |
| Region of residence | Lisboa e Vale do Tejo | 106 (33.3%) | 38 (34.5%) | ref | ||
| Alentejo e Algarve | 32 (10.1%) | 9 (8.2%) | 0.70 (0.29.Çô1.67) | 0.42 | .Çô | |
| Centro | 63 (19.8%) | 17 (15.5%) | 0.66 (0.33.Çô1.31) | 0.236 | .Çô | |
| A.ºores | 12 (3.8%) | 4 (3.6%) | 0.90 (0.25.Çô3.17) | 0.863 | .Çô | |
| Madeira | 10 (3.1%) | 6 (5.5%) | 2.68 (0.71.Çô10.11) | 0.144 | .Çô | |
| Norte | 95 (29.9%) | 36 (32.7%) | 1.09 (0.62.Çô1.94) | 0.764 | .Çô | |
| Socioeconomic | ||||||
| Educational level | 3rd cycle of basic education or less | 132 (42.0%) | 63 (56.3%) | ref | ||
| High school | 80 (25.5%) | 25 (22.3%) | 0.50 (0.28.Çô0.89) | 0.019 | 20.3% | |
| Post-secondary education or more | 102 (32.5%) | 24 (21.4%) | 0.34 (0.19.Çô0.60) | <0.001 | 38.7% | |
| Occupation | Employed | 158 (51.0%) | 44 (40.0%) | ref | ||
| Student | 54 (17.4%) | 13 (11.8%) | 0.82 (0.4.Çô1.68) | 0.59 | .Çô | |
| Domestic | 10 (3.2%) | 5 (4.5%) | 2.59 (0.72.Çô9.39) | 0.147 | .Çô | |
| Looking for a first job or Unemployed | 29 (9.4%) | 19 (17.3%) | 4.92 (2.12.Çô11.42) | <0.001 | 26.8% | |
| Retired | 59 (19.0%) | 29 (26.4%) | 2.51 (1.35.Çô4.65) | 0.004 | 22.3% | |
| Income | <340 Euros | 16 (7.0%) | 11 (12.6%) | ref | ||
| 340.Çô618 Euros | 46 (20.1%) | 26 (29.9%) | 0.59 (0.18.Çô1.98) | 0.393 | .Çô | |
| 619.Çô1531 Euros | 92 (40.2%) | 31 (35.6%) | 0.23 (0.07.Çô0.72) | 0.012 | 57.4% | |
| 1532.Çô3522 Euros | 59 (25.8%) | 17 (19.5%) | 0.18 (0.06.Çô0.61) | 0.006 | 54.0% | |
| >3523 Euros | 16 (7.0%) | 2 (2.3%) | 0.07 (0.01.Çô0.40) | 0.003 | 48.1% | |
| Risk and clinical factors | ||||||
| Body mass index (BMI) | Normal weight | 138 (44.5%) | 40 (37.4%) | ref | ||
| Underweight BMI (kg/m2) <18.50 | 10 (3.2%) | 3 (2.8%) | 1.05 (0.26.Çô4.27) | 0.946 | .Çô | |
| Pre.Çôobesity BMI (kg/m2) .ëÑ25.0 and <30.0 | 115 (37.1%) | 38 (35.5%) | 1.21 (0.71.Çô2.06) | 0.487 | .Çô | |
| Obesity BMI (kg/m2) .ëÑ30.0 | 47 (15.2%) | 26 (24.3%) | 3.03 (1.53.Çô6.00) | 0.001 | 23.5% | |
| Body mass index (BMI), kg/m2 | 25.2 (22.1.Çô28.3)a | 26.3 (23.5.Çô29.7)a | 1.09 (1.03.Çô1.14) | 0.001 | .Çô | |
| Smoking habits | Current smoker | 49 (15.6%) | 18 (15.9%) | ref | ||
| Former smokers | 67 (21.3%) | 19 (16.8%) | 0.68 (0.31.Çô1.50) | 0.34 | .Çô | |
| Non.Çôsmoker | 199 (63.2%) | 76 (67.3%) | 1.06 (0.56.Çô2.03) | 0.851 | .Çô | |
| Having rhinitis symptoms | No | 118 (43.2%) | 25 (22.9%) | ref | <0.001 | |
| At least 1 or 2 days a week | 155 (56.8%) | 84 (77.1%) | 4.40 (2.56.Çô7.58) | 65.9% | ||
| Allergies as a trigger for the crisis | No | 72 (22.0%) | 29 (25.2%) | ref | ||
| Yes | 255 (78.0%) | 86 (74.8%) | 0.76 (0.44.Çô1.29) | 0.305 | .Çô | |
| Clinical care | ||||||
| Family doctor | Having | 262 (81.6%) | 97 (85.1%) | ref | 0.236 | |
| Not having | 59 (18.4%) | 17 (14.9%) | 0.69 (0.37.Çô1.28) | .Çô | ||
| Assistant physician to solve asthma problems | Family doctor/General clinic | 151 (46.5%) | 54 (47.8%) | ref | ||
| Pneumologist | 48 (14.8%) | 18 (15.9%) | 1.08 (0.55.Çô2.11) | 0.827 | .Çô | |
| Allergologist | 78 (24%) | 21 (18.6%) | 0.66 (0.36.Çô1.21) | 0.178 | .Çô | |
| Emergency Care | 32 (9.8%) | 15 (13.3%) | 1.59 (0.73.Çô3.42) | 0.241 | .Çô | |
| Other | 16 (4.9%) | 5 (4.4%) | 0.82 (0.27.Çô2.47) | 0.72 | .Çô | |
| Taking the seasonal flu vaccine | Yes | 130 (40.6%) | 53 (46.1%) | ref | 0.137 | |
| No | 190 (59.4%) | 62 (53.9%) | 0.70 (0.44.Çô1.12) | .Çô | ||
| Use the Peak Flow Metre | Never | 264 (88.6%) | 96 (91.4%) | ref | 0.259 | |
| At least once a month or at doctor Appointment/when have symptoms | 34 (11.4%) | 9 (8.6%) | 0.63 (0.28.Çô1.41) | .Çô | ||
| Perform lung function tests (past 12 months) | At least every six months or at all Doctor appointment | 105 (43.8%) | 36 (40.9%) | ref | ||
| Only once | 116 (48.3%) | 46 (52.3%) | 1.26 (0.73.Çô2.18) | 0.410 | .Çô | |
| Never | 19 (7.9%) | 6 (6.8%) | 0.89 (0.31.Çô2.52) | 0.819 | .Çô | |
| Have training to use the inhaler properly | Yes | 253 (79.8%) | 90 (81.1%) | ref | 0.679 | |
| No | 64 (20.2%) | 21 (18.9%) | 0.88 (0.49.Çô1.58) | .Çô | ||
| Patients having a written asthma action plan | Yes | 77 (24.9%) | 23 (21.3%) | ref | 0.281 | |
| No | 232 (75.1%) | 85 (78.7%) | 1.36 (0.78.Çô2.37) | .Çô | ||
| Treatment | ||||||
| Forgetting to take medication | Yes | 52 (33.3%) | 19 (26.8%) | ref | 0.113 | |
| No | 104 (66.7%) | 52 (73.2%) | 1.74 (0.88.Çô3.44) | .Çô | ||
| Under inhaled corticosteroid | Using | 63 (31%) | 31 (42.5%) | ref | 0.009 | |
| Used in the past or never used | 140 (69%) | 42 (57.5%) | 0.44 (0.24.Çô0.82) | 46.7% | ||
| Number of asthma medication | None or one medication | 162 (49.5%) | 49 (42.6%) | ref | 0.065 | |
| Two or more medications | 165 (50.5%) | 66 (57.4%) | 1.54 (0.97.Çô2.43) | .Çô | ||
CI: Confidence Interval; ref: reference.
Data shown in this table exclude missing values and answers .Ç£do not know/do not respond.Ç¥.
Multivariable analysis for uncontrolled asthma with sequential dimensions adjustments.
| Determinants | Adjusted to A | Adjusted to A+B | Adjusted to A+B+C | ||||
|---|---|---|---|---|---|---|---|
| Adjusted OR (95% IC) | P value | Adjusted OR (95% IC) | P value | Adjusted OR (95% IC) | p value | ||
| Demographic (A) | |||||||
| Age | 1.02 (1.00.Çô1.03) | 0.01 | / | / | |||
| Sex | Male | Ref | / | / | |||
| Female | 1.77 (1.08.Çô2.90) | 0.023 | |||||
| Socioeconomic (B) | |||||||
| Educational level | 3rd cycle of basic education or less | Ref | / | ||||
| High school | 0.61 (0.31.Çô1.22) | 0.16 | 0.67 (0.33.Çô1.34) | 0.255 | |||
| Post-secondary education or more | 0.38 (0.21.Çô0.72) | 0.003 | 0.47 (0.24.Çô0.90) | 0.023 | |||
| Occupation | Employed | Ref | ND | ||||
| Student | 0.92 (0.41.Çô2.06) | 0.831 | 0.75 (0.32.Çô1.74) | 0.498 | |||
| Domestic | 1.98 (0.51.Çô7.61) | 0.322 | 5.09 (0.54.Çô47.96) | 0.155 | |||
| looking for a first job or unemployed | 4.55 (1.95.Çô10.63) | <0.001 | 6.05 (2.21.Çô16.57) | <0.001 | |||
| Retired | 2.11 (0.90.Çô4.93) | 0.084 | 1.71 (0.79.Çô3.69) | 0.171 | |||
| Income | <340 Euros | Ref | / | ||||
| 340.Çô618 Euros | 0.78 (0.23.Çô2.68) | 0.692 | 0.73 (0.19.Çô2.90) | 0.66 | |||
| 619.Çô1531 Euros | 0.35 (0.11.Çô1.15) | 0.084 | 0.36 (0.09.Çô1.40) | 0.14 | |||
| 1532.Çô3522 Euros | 0.27 (0.08.Çô0.93) | 0.038 | 0.32 (0.08.Çô1.31) | 0.112 | |||
| >3523 | 0.13 (0.02.Çô0.87) | 0.035 | 0.12 (0.02.Çô0.88) | 0.037 | |||
| Risk and clinical factors (C) | |||||||
| Body mass index (BMI) | 1.07 (1.02.Çô1.13) | 0.008 | 1.07 (1.01.Çô1.13) | 0.017 | 1.09 (1.02.Çô1.16) | 0.012 | |
| Having rhinitis symptoms | No | Ref | |||||
| at least 1 or 2 days a week | 4.63 (2.64.Çô8.11) | <0.001 | 4.49 (2.49.Çô8.10) | <0.001 | 4.71 (2.52.Çô8.81) | <0.001 | |
| Treatment (E) | |||||||
| Under inhaled corticosteroid | Using | Ref | / | ||||
| used in the past or never used | 0.48 (0.25.Çô0.89) | 0.02 | 0.49 (0.26.Çô0.94) | 0.032 | |||
CI: Confidence Interval; Ref: Reference.
ND: not determinate because A+B adjustment is occupation.
/: not represented because p values >0.05.
A: Age and Sex; R2 Cox=0.040 and Nagelkerke=0.055.
A+B: Occupation; R2 Cox=0.074 and Nagelkerke 0.101.
A+B+C: Occupation, IMC and Having rhinitis symptoms; R2 Cox=0.201 and Nagelkerke=0.272.
The following adjusted models A+B+C+D and A+B+C+D+E didn.ÇÖt add any of the determinants considered in Clinical Care or Treatment and are not presented in this table.
Individuals over age of 65 had 2.70 (95% CI: 1.21.Çô5.99) times higher odds for having uncontrolled asthma compared to the youngest group analyzed (16.Çô25 years) and PAR% value of 19.3%. It appears that there is a progressive increase; with each year of age the chance of uncontrolled asthma increases by 2% and female gender increases the odds of uncontrolled asthma by 1.87 (95% CI: 1.15.Çô3.04). However socioeconomic determinants (occupation) seems to attenuate these associations.
Regarding socioeconomic determinants: the increase in education level decreases the odds of having uncontrolled asthma (OR 0.5 high school level or more); higher income decreases the odds of uncontrolled asthma; in relation to occupation, subjects looking for a first job or unemployed had 4.92 higher odds for uncontrolled asthma scores (95% CI: 2.12.Çô11.42) and being retired is associated with 2.51 odds (95% CI: 1.35.Çô4.65) for uncontrolled asthma, when compared with employed asthmatics. The protective role of educational level and income seems to be mediated by risk and clinical factors (BMI and having rhinitis symptoms). Participants looking for a first job or who were unemployed had PAR% value of 26.8% and were much more likely to have uncontrolled asthma regardless of the various adjusted dimensions.
For each kg/m2 increment of BMI the chance of uncontrolled asthma increased 9%, this association remained significant even when dimensions adjustments were considered. PAR% of uncontrolled asthma associated with obesity was 23.5%.
Nasal symptoms were the symptoms identified as occurring almost every day in the previous four weeks (Fig. 1). Having rhinitis symptoms increases the odds of uncontrolled asthma by 4.4 (95% CI: 2.56.Çô7.58). These associations remained significant regardless of the adjustments considered. The PAR% of uncontrolled asthma associated with rhinitis symptoms was 65.9%.
Clinical factors, such as smoking habits and having allergies as a trigger for asthma crisis, were not significantly associated with uncontrolled asthma. Also, in this analysis, the determinants related to clinical care did not demonstrate significant associations. Of the treatment determinants evaluated (forgetting to take medication, the number of medications used for asthma and the use of inhaled corticosteroids) individuals not using inhaled corticosteroids had significantly lower odds for uncontrolled asthma (OR 0.44 95% CI: 0.24.Çô0.82), but it seems that the effect is mediated by the risk factors and the clinical aspects considered (BMI and having rhinitis symptoms).
DiscussionThis study showed that 64.8% subjects had their asthma controlled, and 35.2% of them had uncontrolled asthma, according to the evaluation of a questionnaire for asthma control purpose, the ACT.
Evaluation of asthma control has been assessed in diverse countries worldwide and despite the different measurement tools used, a sub-optimal level of control has been revealed.
Results from a multinational study involving twelve countries of Europe (n=3.123), indicate a similar overall percentage (38.5) for uncontrolled asthma, with the lowest levels of asthma control found in Hungary, The Netherlands and France.7 Higher levels of uncontrolled asthma (71.3%) were found in surveyed African countries (Algeria, Morocco, and Tunisia),19 USA (71%),20 Asia-Pacific countries (92.4%)21 and Latin America (93%).22
From a national perspective, the Portuguese National Asthma Survey using Control of Allergic Rhinitis and Asthma Test (CARAT), reported 43.1% of uncontrolled asthma in Portuguese population.13 In 2007, a study in a central hospital in Lisbon using the ACT questionnaire in the context of allergology consultation found that 23.3% of the respondents presented ACT values .ëñ1911 and a study carried out in Portuguese pharmacies, with the application of the same questionnaire to evaluate the control, revealed even higher scores for poor asthma control (61.2%).23
In this study it was possible to observe that the level of control assessed can differ with the asthma control tool utilized. The different methodologies that can be used in epidemiological studies such as in clinical practice for control assessment [e.g., GINA based symptom control tool1; CARAT24; Asthma Control Questionnaire (ACQ)25; ACT16] can make it more difficult to compare results between studies. Additionally, there are different approaches used to gather information (e.g. telephone interview, online questionnaire, face-to-face interview), several methods used to assess asthma control level and different situations in different health systems, literacy and social barriers among the different countries; all of which should be taken into consideration when comparing results from different studies.
Most of the adult patients interviewed overestimated their disease control revealing a discrepancy between patient perception of asthma control and the objective measurement, since 93.9% of the individuals perceived their asthma with some level of control. It has been shown that most of these patients can tolerate their symptoms and lifestyle limitations as inevitable consequences of the disease.26 The self-perception and expectations about asthma disease may result in misinterpretations of control level.26
In terms of demographic factors, our results indicate that uncontrolled asthma was more frequently associated with older age and with female sex.
Asthma disease in the elderly population is of special concern since most of patients are polypathological and polymedicated, which can have a negative impact on disease management.27 Besides, there are often more difficulties in the execution of proper inhaler techniques.27,28 K.ñmpe et al.29 stated that uncontrolled asthma was more common in older groups (.ëÑ65years); Mendes et al.,23 using a Portuguese population, found that 69% of elderly (>70 years) had uncontrolled asthma.
Our results observing the association between sex and poor asthma control, are in agreement with other studies,23,29,30 with females associated with poor scores in asthma control. Usually women perceive asthma differently from men, reporting higher levels of symptoms and more limitations in day-to-day life.31,32 In addition, women have been associated with a higher incidence of later asthma onset and a higher prevalence of non-allergic asthma phenotype, favouring a less effective response to corticosteroid treatments.1,33,34
In this study, socioeconomic determinants, especially occupation have a considerable association with poor control score of asthma as well as greater influence on other important determinants. The PAR% for being looking for a first job or being unemployed was 26.8%, suggesting a very significant contribution for poor asthma control. Moreover studies identified that the severity of asthma,35 the frequency of asthma symptoms and night awakenings because of asthma can contribute to work disability and unemployment.36
The inverse association between income37 and education29 with poor controlled asthma outcomes has been documented. The PAR% register for having at least post-secondary education was 38.7% suggesting a contribution for preventing worse levels of control in asthma. The same positive contribution was verified for higher levels of income.
Individuals with higher levels of education can have a better understanding of the prescribed therapeutics and may have a greater tendency to adhere to treatment regime.29 Furthermore, there are work environments associated with trigger agents exposure (e.g., diisocyanates, acrylates or cleaning agents)38 to which individuals with higher educational levels are less likely to be exposed.29
Although only 8.8% of the individuals considered that the price of asthma treatments influenced taking medication according medical indications, asthma expenses related to medical treatments are usually high which can be a significant burden for low-income families,39,40 and in turn can compromise treatment adherence.
Higher BMI was shown to be associated with uncontrolled levels in asthma, as stated above: Lessard et al.41 found that regardless of a similar perception of symptoms, obese individuals had poorer asthma control than non-obese asthmatics; Lavoie et al.42 in a 382 adults study found that patients with higher BMI had poor asthma control (scored higher in ACQuestionnaire) independent of their age, sex and asthma severity; with an odds ratio of 2.99 (95% CI 1.14.Çô7.08) Ferreira et al.11 reported that obesity is a factor associated with non-control of asthma.
Obesity has a negative impact on overall breathing mechanisms,43 has been reported as a risk factor for the development of asthma and for increasing exacerbations and hospitalizations.44 The pro-inflammatory characteristics of obesity can induce more difficulties in asthma control, secondary to a reduction in the response to corticosteroids.45
Weight loss has been shown to improve lung function parameters in individuals with obesity and better scores of disease control in asthma patients.44,46 In this way, nutritional education promoting diet modification and weight loss in obese asthmatic patients must be encouraged.47 According to our results a considerable number of individuals (PAR%=23.5%) with uncontrolled asthma could be spared if obesity was not present.
Findings from studies11,48.Çô50 conclude that rhinitis is an important contributor to a non-optimal asthma control level, as also was supported by our results. Bousquet et al.51 in a multivariate analysis found that patients with simultaneous asthma and allergic rhinitis are associated with higher levels of acute exacerbations (OR 1.35, 95% CI: 1.03.Çô1.77) with more emergency visits (OR 2.35, 95% CI: 1.12.Çô4.80).
The PAR% register for having rhinitis symptoms in our study (65.9%) suggests that controlling nasal symptoms could contribute positively to preventing worse levels of control in asthma disease. Consequently, it is important to optimize the treatment of both diseases in patients with a concomitant diagnosis.52
The presence of smoking habits was not observed to be a determinant associated with poor asthma control in our study, as already stated.30,53 In this regard, other studies also did not identify smoking as an independent risk factor for uncontrolled asthma11 or for decline in lung function in asthmatic patients54 which could be related to a healthy smoker effect,55 underestimating the effect of tobacco at pulmonary level.
Nor was an association found with determinants of poor asthma control that have been reported in other studies, such as patients having a written asthma action plan56 and therapeutic compliance. Although a higher number of individuals have reported allergies as a trigger for asthma crises (78%), a significant association with uncontrolled asthma was not detected, which is similar to that reported by other research.11,53
Lung function assessment with peak flow metre or lung function tests were not significant determinants for asthma control in our study. However minimizing airflow limitation is one of the main goals for these patients and the importance of these exams in diagnosis and follow-up of asthma has been stressed.1
The use of inhaled corticosteroid plays an important role in an asthma treatment plan.1 Rabe et al.15 reported that the use of inhaled corticosteroids was low even among patients with severe persistent symptoms. In our study, the use of these medications was associated with lower ACT scores, which represents worse control outcomes. This association with the level of control could be explained by the relation of treatment plan with disease severity. For example, patients with mild asthma degree and well controlled symptoms might only be at step 1 of treatment plan and only using a reliever inhaler as needed, as was defined before 2019 GINA guidelines.1 However, severe asthma patients may present corticosteroid resistance,57 of which the molecular pathophysiology is beginning to be understood,58 leading to the possibility of development of new treatments for these patients.
Strength and limitationsThere are some limitations to this study that should be considered: (1) it is a cross-sectional study and cause-effect relationships cannot be established; (2) all the data was reported by respondents and could not be clinically confirmed; (3) only patients with active asthma were questioned, excluding undiagnosed individuals and patients without the presence of symptoms and without medication in the last year; (4) this study did not inquire about comorbidities, such as: other respiratory disease which have been associated to asthma control [e.g. Chronic Obstructive Pulmonary Disease (COPD), obstructive sleep apnoea, gastroesophageal reflux disease (GERD), or psychological aspects]59; (5) the degree of asthma severity, according to GINA guidelines, was not assessed.
A strength of this study was the identification of active asthmatic patients from the general population without referral bias. It was also important to evaluate different methods of accessing data from the level of asthma control and the analysis of multiple factors and dimensions that could have influenced the level of control of this important chronic disease.
ConclusionsIn this study uncontrolled asthma was associated with several factors: age, sex, educational level, occupation, income, BMI, having rhinitis symptoms and use of inhaled corticosteroids as part of a treatment plan. Some of these factors remain associated after full adjustment, notably occupation, BMI and having rhinitis symptoms, which are important modifiable factors.
The values of PAR% for unemployment, obesity and rhinitis symptoms were relatively high (respectively 26.8%, 23.5% and 65.9%). Since these are modifiable factors from the point of view of public health and health planning, intervention strategies more directed to these concerns can promote benefits for asthmatic patients and consequently for other health problems. Similarly, their identification by physicians could contribute to a better clinical approach to asthma disease.
In future research there should longitudinal studies to confirm these associations and other comorbidities, such as COPD or GERD and enquiries made about physiological factors. It is also important to standardize a tool for asthma control evaluation that can be used in epidemiological studies and that can provide reliable comparison between studies.
FundingThis study was supported by a grant from the General Directorate of Health (DGS) of the Portuguese Health Ministry and by a scientific grant from AstraZeneca Foundation. The supporters did not have any role in the design and conduct of the study, nor in the collection, management, analysis, and interpretation of the data, or in the preparation, review or approval of the article.
Conflicts of interestThe authors have no conflicts of interest to declare.
We acknowledge the help of Filipe Le.úo Miranda, Augusto Almeida, Rui Sim..es, Fernando Martins e Ana Sofia Andrade for valuable inputs, project management, sampling and data collection instruments and algorithms, and previous data analysis. We are also grateful to F.ítima Rodrigues for careful review of the manuscript and to all who gave support and participated, making this study possible.