Acquired subglottic stenosis (SGS) occurs in 1–2% of children with a history of intubation. An alternative treatment is endoscopic dilation with rigid dilators.
Material and methodsSeventy-four patients with SGS grade I to III were treated between 2003 and 2017. Dilations were performed with Hegar-type rigid dilators every 2–3 weeks.
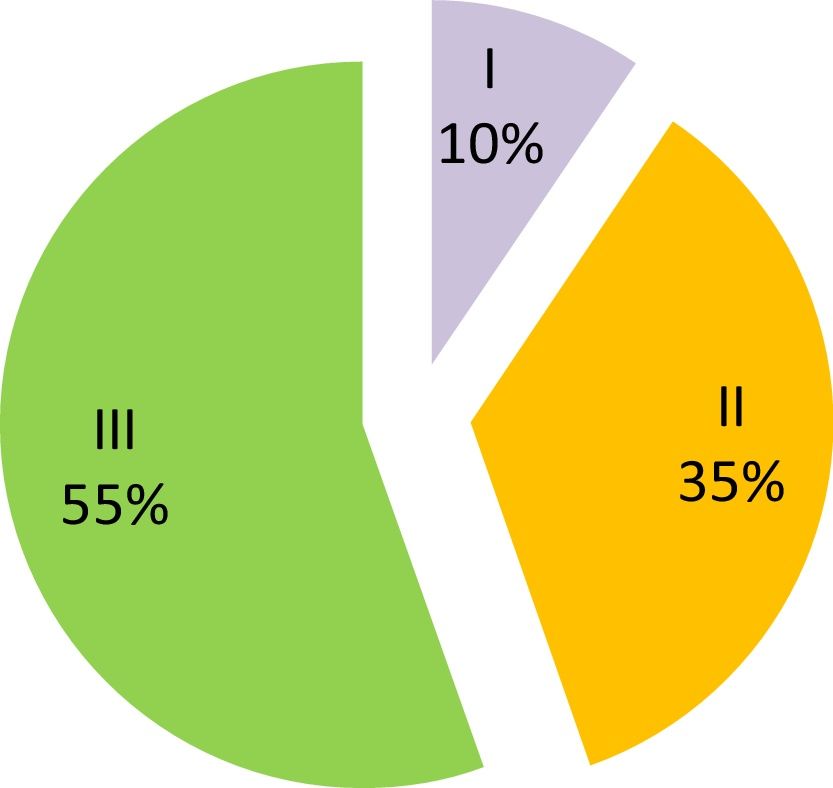
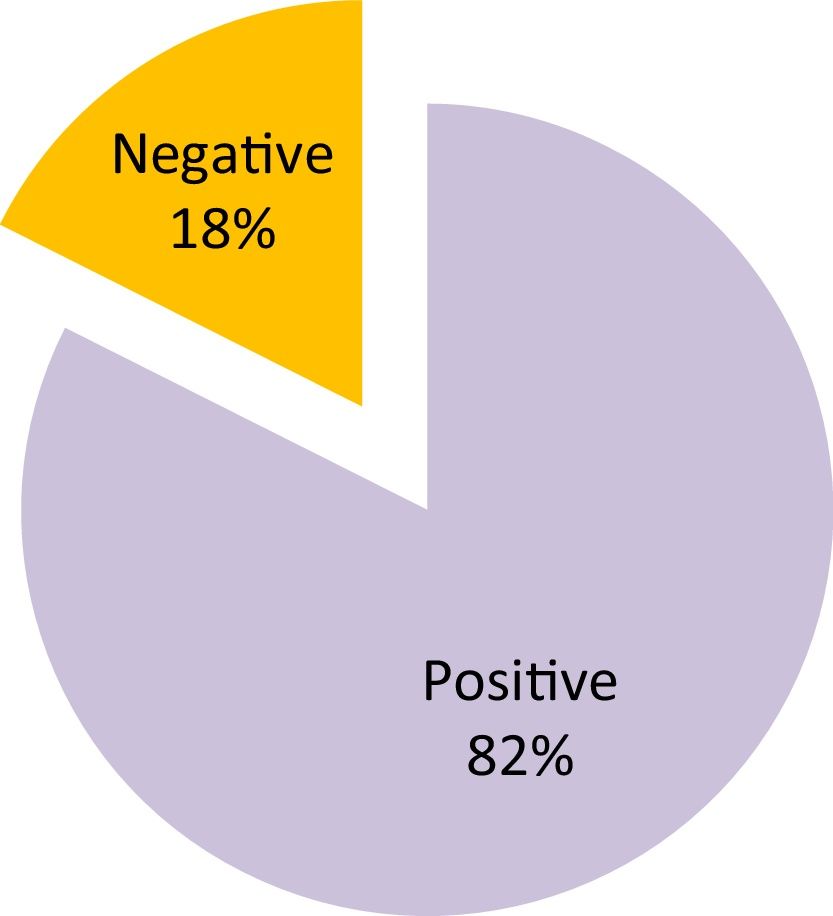
ResultsEighty-two percentage of patients responded to the treatment. 10% presented SGS grade I, 35% grade II and 55% grade III. Previous intubation time in successful cases was 12.4 days and it was 32 days in those that failed (p=0.02). The average number of dilations was 3.23 in the group that responded and 2.98 for those that did not respond (p=0.51). The presence of tracheostomy reduced the effectiveness of the treatment (p=0.002). The average follow-up was 43.5 months.
ConclusionThe use of rigid dilators under endoscopic control is an effective minimally invasive method for treating patients with SGS grades I to III. Previous intubation time and the presence of tracheostomy were identified as poor prognostic factors.
Subglottic stenosis (SGS) is the narrowing or reduction of the airway lumen at subglottis level affecting the cricoid cartilage. This can be congenital or acquired. More than 90% of cases of SGS are secondary to endotracheal intubation.1,2
Until the 1980s, endoscopic dilations were most often used to treat subglottic stenosis. However, surgical reconstruction began to boom after the publications of Fearon and Cotton.3 Open surgery includes laryngotracheal reconstruction with costal cartilage and partial cricotracheal resection, both with defined indications and successful results.3–5 These are invasive and time-consuming surgeries, with hospitalization in intensive care units, morbidity and even mortality. Some of the patients will even require post-surgical endoscopic treatments to improve results.6
Endoscopic treatments include dilations with pneumatic balloons or rigid dilators, laser incisions or cold scalpel.6 Endoscopic balloon dilations have greater acceptance due to their advantage expanding the stenotic area through their measured and controlled radial pressure.7 Rigid dilators were sidelined because the shear forces applied in the stenotic sector hypothetically cause mucosal injury, resulting in fibrosis and worsening of the disease. Some authors have reported encouraging results with rigid metal dilators with the benefit of lowering costs by using reusable instruments over a long period of time.8
Endoscopic dilations were indicated for stenoses grade I and II and in those SGS recurrences in post-surgical patients with laryngotracheal reconstructions.9 Currently, patients with SGS grade III are also candidates for dilations.2
The aim of this study is to evaluate the results of treatment with rigid dilators and identify factors of prognostic value.
Materials and methodsThis is an observational, analytical – descriptive, retrospective and longitudinal analysis of clinical histories of patients diagnosed with acquired subglottic stenosis (SGS) treated with dilations in a 14-year period between March 2003 and March 2017 in the Airway Department of “Hospital de Niños de la Santísima Trinidad‿.
Population: Patients with a diagnosis of SGS whose initial treatment was dilations are the subject of this analysis.
We included all patients with SGS secondary to endotracheal intubation and who were initially treated with endoscopic dilations. We excluded patients with congenital SGS, those with previous laryngeal surgery and patients with SGS grade IV.
Subglottic stenosis diagnosis was made under general anesthesia using a rigid bronchoscope zero degree lens with an appropriate caliber for the patient's age. The stenosis caliber was measured with Myer–Cotton technique and dilation was performed with Hegar-type rigid dilators with increasing diameters until reaching the patient's corresponding diameter was reached.10 The largest caliber dilator was left in the narrow zone for a period of 2min or until saturation fell below 92%. The result of the expansion was then evaluated with the same technique. Four operators performed all procedures.
The dilations were repeated every 2 or 3 weeks depending on the patient's clinical condition.
The patients were treated as outpatients and were discharged 6–8h after endoscopic dilation. In all cases, empirical treatment for gastroesophageal reflux with proton pump inhibitors was indicated.
We evaluated the age (months), previous intubation time that produced the lesion (days), diagnostic interval (days from extubation to SGS diagnosis), degree of stenosis according to Myer and Cotton classification (grade I: 0–50%, grade II: 51–70%, grade III: 71–99% and grade IV: no detectable lumen), presence of tracheostomy prior to treatment, airway associated pathologies (larynx, vocal cords, trachea and bronchi), response to treatment, decannulation in tracheostomized patients and follow-up (months).
A good or “positive‿ response to treatment was considered when an adequate and stable caliber of the airway was achieved (less than 30% stenosis) and the patient remained asymptomatic and without limitations in normal activity for his or her age. Otherwise it was considered a failure or a “negative‿ result.
Statistical methodologyAn Excel-like database was created for statistical processing using data collected from clinical histories. Central and dispersion measures were calculated for the descriptive statistics of quantitative variables, and percentages for each category were calculated for the categorical variables. Variable comparison according to “positive‿ and “negative‿ results was accompanied by a Wilcoxon inferential statistical test in the case of mean comparison and a chi-square for categorical results. The level of significance used was 0.05 and it was carried out with the SPSS program.
ResultsDuring the period analyzed a total of n=74 children with acquired SGS diagnosis were treated. Fifty-five percent of the patients were male and the remaining 45 percent were female. The mean age at diagnosis was 19.4 months (SD=30.8), ranging from 1 month to 12 years.
As for the initial diagnosis that required patient intubation, the most frequent were bronchiolitis 31%, heart diseases 13.5%, pneumonias 10%, prematurity 9% and respiratory distress syndrome due to various causes. Eight percent of the patients had an associated diagnosis of Down Syndrome.
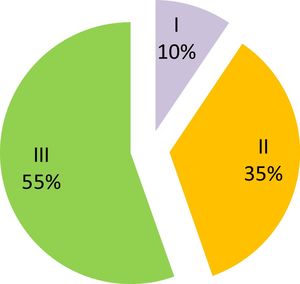
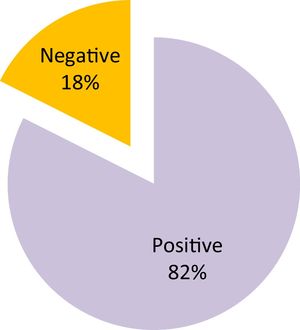
Mean intubation time was 16.2 days (SD=18.8) and stenosis was grade I in 10%, grade II in 35% and grade III in 55% of cases (Fig. 1). Response to treatment was “positive‿ in 82% of cases and “negative‿ in the remaining 18% (Fig. 2).
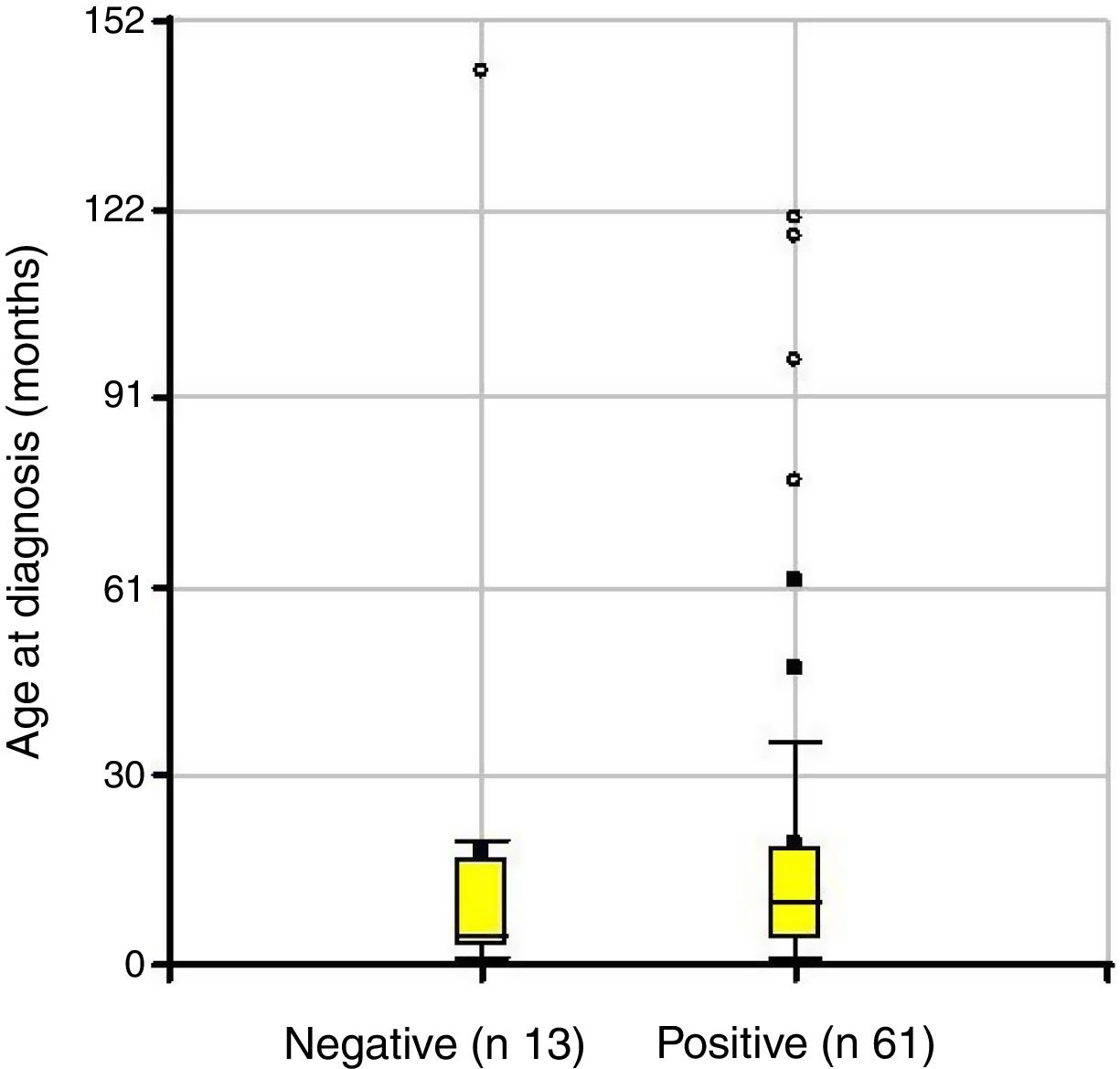
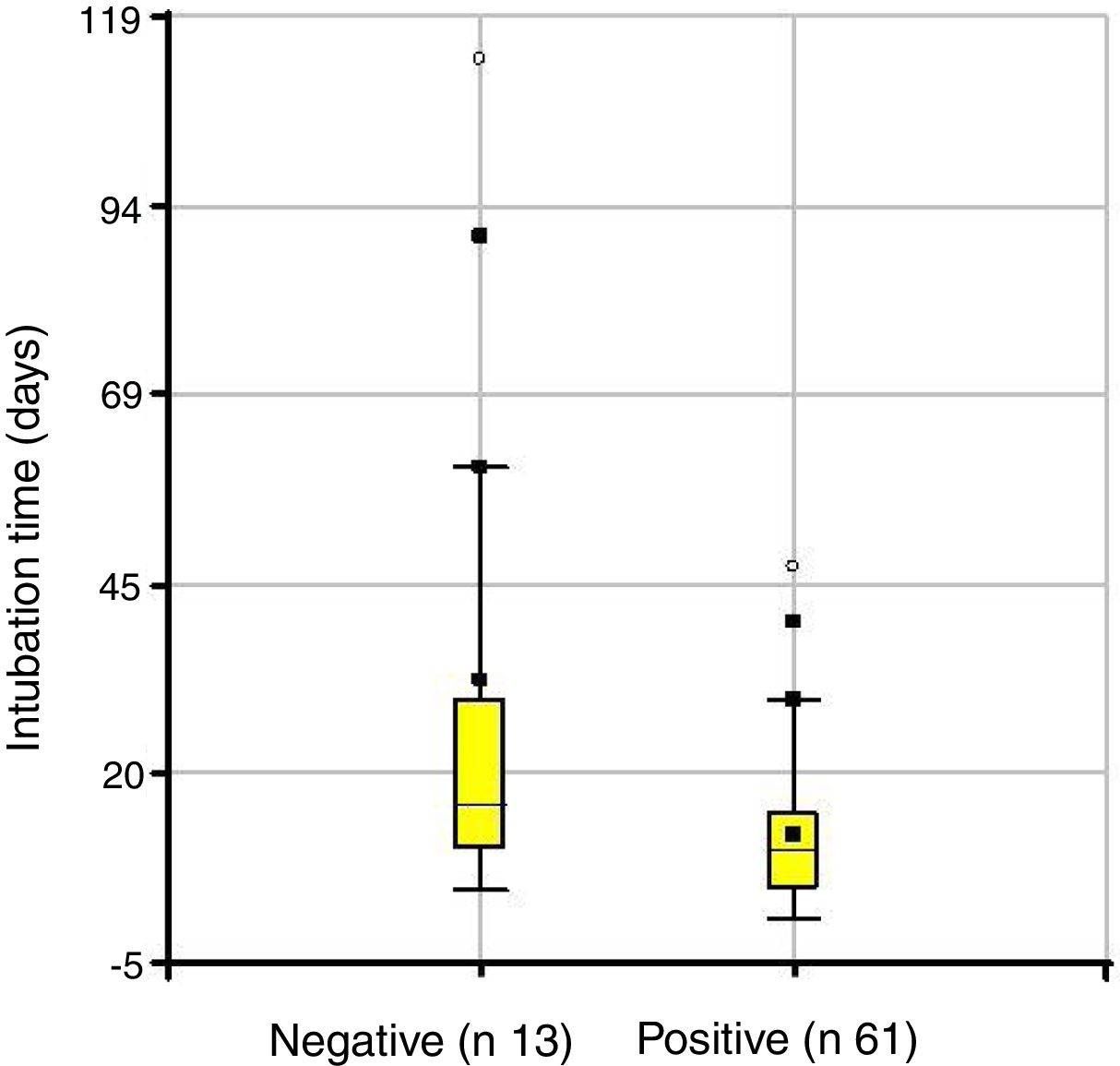
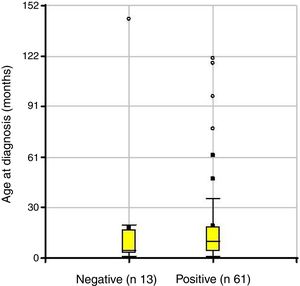
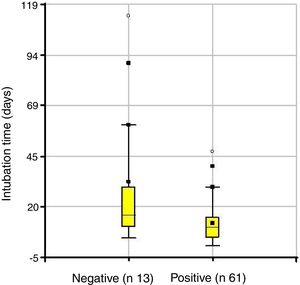
Depending on the treatment outcome, the mean age at diagnosis was 18 months for those with a positive result and 19.7 months for those who did not respond (p=not significant) (Fig. 3). The intubation time in positive cases was 12.4 days and 32 days in the group where treatment failed (p=0.0231) (Fig. 4).
The average number of dilations was 3.23 in the respondent group and 2.98 for non-responders (p=0.51). The diagnostic interval in patients with positive results was 836.7 days (r: 1–31.360), of which 73% exceeded 30 days of extubation. In patients with negative results, this interval was 97.6 days (r: 1–540 days).
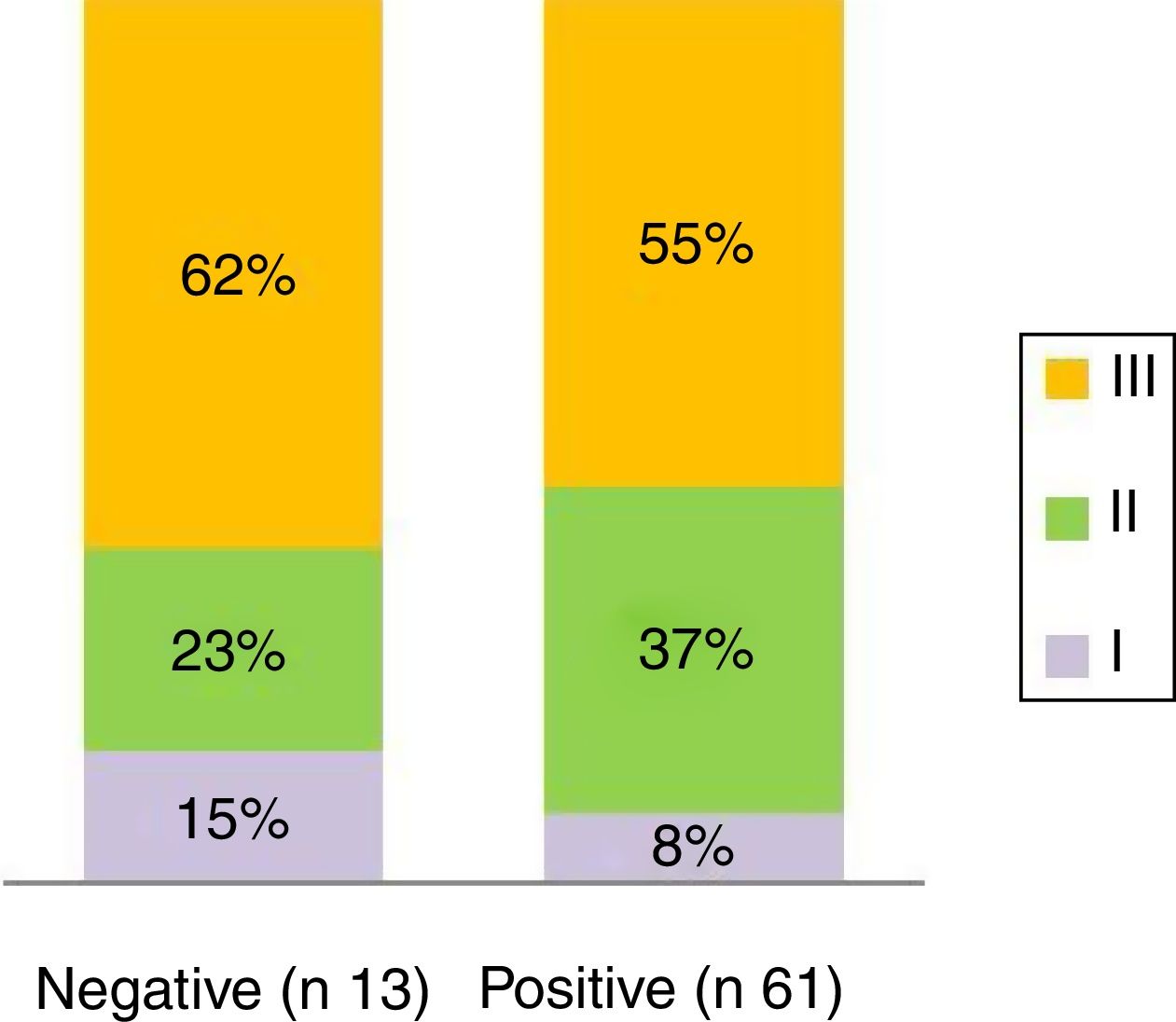
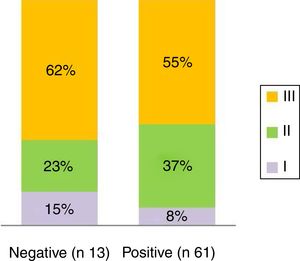
From the analysis of stenosis degree it is concluded that the majority of patients in both groups presented grade III: 62% of the negatives and 55% of the positives, while the percentages with stenosis grade II were 23% and 37% respectively (Fig. 5).
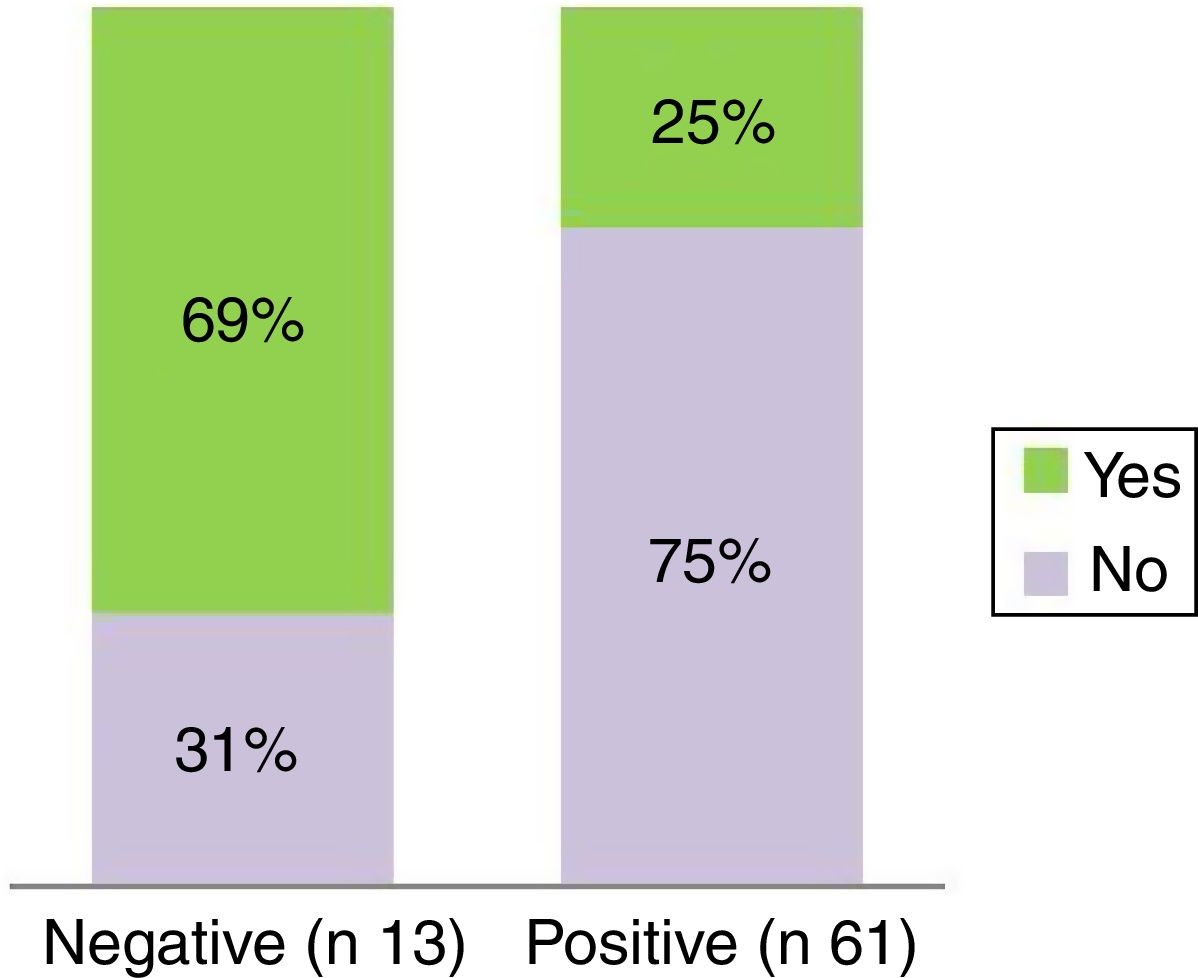
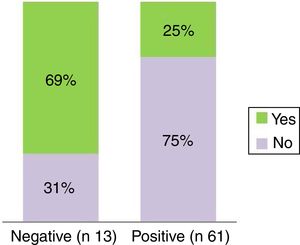
Nine patients who did not respond to dilation treatment had previous tracheostomy (p=0.0021) (Fig. 6). Among the reasons for performing tracheostomy are: pulmonary pathology (broncopulmonary dysplasia, pneumonia, bronchiolitis), vocal cord paralysis, and prolonged intubation in neonatal period.
Eighteen patients were diagnosed with associated airway pathology including unilateral vocal cord paralysis, bilateral vocal cord paralysis, laryngomalacia, and laryngotracheomalacia among others. Fourteen patients presented airway comorbidities in the responding group and 4 patients in the group with poor outcome. Statistical analysis indicated that the difference is not significant (p=0.5508). Patients with glottic or tracheal stenosis were not included.
Among the 13 patients who did not respond to treatment with dilations, eight received open surgical treatment with good results in six of them. Technically, surgery did not present complications related to previous treatment. The remaining patients are awaiting surgical resolution. The characteristics of the patients with negative results are described in Table 1.
Characteristics of patients with negative results in relation to diagnosis.
| Diagnosis | Comorbility | ESG | Traqueostomy |
|---|---|---|---|
| BPD | 95 | Yes | |
| Down syndrome+BQL | Laryngomalacia | 60 | No |
| BPD | 80 | Yes | |
| BQL | 70 | No | |
| Pneumonia | 90 | Yes | |
| Prematurity | Laryngomalacia | 80 | Yes |
| BQL | 90 | Yes | |
| Pneumonia | 50 | No | |
| Cardiopaty | VC paralysis | 60 | Yes |
| Pneumonia | 80 | Yes | |
| Cardiopaty | VC paralysis | 85 | Yes |
| Prematurity | 50 | No | |
| BQL | 90 | Yes |
BPD: bronchopulmonary dysplasia. BQL: bronchiolitis. VC: Vocal cord. ESG: subglottic stenosis, obstruction percentage.
The average follow-up for all patients was 43.5 months starting from the last bronchoscopy to the last office control (r: 1–86 months).
Four patients died. The causes were: tracheostomy cannula accident, sepsis, nosocomial pneumonia and complex congenital heart disease.
There were no complications attributable to the endoscopic dilation procedure with rigid dilators.
DiscussionSubglottic stenosis is the most severe consequence of endotracheal intubation in children.11 A simple, effective, minimally invasive therapy could be of great benefit for the treatment of this pathology. The option of dilating the narrow airway is one of them. Balloon dilations have been used with good results.7–10,12,13 However, there are few reports of dilations with rigid or semi-rigid dilators.8,14 Despite the increase in the implementation of treatment with dilations, no selection criteria have yet been established in order to optimize results.15 The success rate of endoscopic treatment has varied according to the reports and the method used, ranging from 60% to 70% by balloon dilations7,13 up to 100% reported with rigid dilations although this includes a large percentage of patients with stenosis grade 1 and a high number of procedures over a long period of time.8
Rigid endoscopic dilation was used on all our SGS diagnosed patients who had airway lumen that allowed a dilator to be introduced. In our case, the result was favorable in 82% of the patients treated. Age and degree of stenosis were not significant factors in predicting response to treatment, consequently it can be stated that it is a therapy applicable to all patients with SGS grade I to III.
When analyzing intubation time we noted that children who responded to treatment had an average of 12.4 days of intubation and in the group that failed it was 32 days (p=0.02). This difference could be due to the fact that longer intubation time would produce a more extensive lesion in length and depth with greater alteration of the cricoid cartilage histology.1
We noted that an average of 3 dilations were enough to determine an outcome in both groups. We consider that other therapeutic alternatives should be considered for those patients who do not show improvement after the third dilation.
The average number of days of diagnostic interval in patients who responded to dilations was higher than in those who failed (836 vs. 97 days), which allows us to suspect that the wound healing period in which the stenosis is found does not define the response to treatment. As mentioned above, 73% of the SGSs that responded had chronic stenosis after more than 30 days of extubation. This may be due to the fact that histological changes are variable and apparently do not depend on time elapsed or severity of the stenosis.11
Bearing in mind that stenosis degree and diagnostic interval did not influence the response unlike intubation time which produced the lesion, we can infer that the most significant factor in predicting the response to dilations could be the depth of histological changes in the airway matrix although we do not have histological analyses to support this hypothesis.1
Another prognostic factor we identified as being associated with a negative response was the presence of tracheostomy. More than half the patients tracheostomized prior to the beginning of endoscopic treatment did not respond. We did not identify any significance in this relationship, since previous intubation times or association of comorbidities were no more frequent in patients with a negative response. Nor did we note lesions associated with endoscopic dilations in patients with tracheostomy.
The presence of airway comorbidities in patients who did not respond to treatment was less than in those with positive results, thus contrary to reports that relate comorbidities with poor response.7 Despite this difference, the analysis was not statistically significant, but we suggest that the presence of associated airway pathology does not contraindicate dilation treatment.
Lang et al. report an average follow-up of 7 months in most papers when conducting a meta-analysis and systematic review in 2013.15 We have a 43 months recurrence-free follow-up of our patients which allows us to assert that this is a treatment with good long-term results. Due to the good results obtained, we have extended the indication of dilations to increasingly severe stenosis since 2003. Among our patients, the highest percentage had stenosis grade 3 with a good response to treatment, which encourages us to implement this method in all patients who have SGS with any lumen allowing dilation.
We conclude that SGS treatment using rigid dilators under endoscopic control is a minimally invasive and effective method. Prolonged intubation time and the presence of tracheostomy were the only factors significantly related to treatment failure. We have not found any factors that contraindicate dilations and dilation with rigid dilators could be indicated as first-line treatment for SGS grade 1–3. We recommend 3 endoscopic dilation procedures and then, in the absence of response, evaluation of other treatment alternatives. Given that the patients who did not respond were subsequently operated on without any difficulty attributable to the dilations, we consider that the initial endoscopic treatment does not interfere with future surgical treatment in the event of failure.
Conflicts of interestThe authors have no conflicts of interest to declare.