Lung cancer is a major global health problem. Several strategies are required to conquer this cancer. Stricter implementations of tobacco control measures are necessary. Early detection programs should be implemented to decrease lung cancer mortality. Although chemotherapy remains a cornerstone of treatment, targeted therapies and immune checkpoint inhibitors improved treatment of metastatic cancers and are hoped to improve outcome of adjuvant and induction therapies. Novel immunotherapy approaches hold great promise. Better understanding of the molecular biology of lung cancer should lead to rational drug design.
Lung cancer is a global health problem. Approximately 2.1 million individuals are diagnosed with lung cancer and 1.8 million die from this cancer each year.1 Lung cancer rates continue to increase on the global level, although the rates are declining among males in some Western countries. Non-small cell lung cancer (NSCLC) makes up about 85% and small cell lung cancer (SCLC) about 15% of lung cancers. Pathological diagnosis is based on histology, immune histology and molecular analysis.2 Lung cancers are currently staged according to the eighth edition of the TNM Classification for Lung Cancer.3,4 Tumor stage is important for prognosis and treatment.3,4 Overall, the five year survival rates are 15–20%. Among patients with NSCLC, these rates reach 90% for stage 1A1 but drop below 10% for stage 4. Among patients with SCLC, the rates are about 30% for limited disease and below 10% for extensive disease.
Treatment of patients with lung cancer requires multidisciplinary co-operation and is based on surgery, radiotherapy, systemic treatments (chemotherapy, targeted therapies, immune checkpoint inhibitors) and supportive care including end-of-life care. Treatment depends on tumor characteristics, tumor stage and patient-related factors. Finally, access to and re-imbursement of novel drugs are becoming an increasing challenge for many countries.
Major diagnostic and therapeutic advances have occurred during the last three decades. Here, the current status of systemic treatment and strategies for conquering lung cancer are described.
Tobacco controlSmoking is by the far the most important risk factor for developing lung cancer. About 80% of lung cancers in Western countries are directly related to smoking. In order to decrease the incidence and mortality rates, therefore, stricter implementation of tobacco control measures is required. These measures are outlined in the WHO Tobacco Free Initiative which includes WHO Framework Convention on Tobacco Control (www.fctc.org) and MPOWER (www.who.int/tobacco/mpower/en/). The single most efficient measure to decrease smoking rates is to raise taxes on tobacco products. Other important measures include smoke-free environment, advertising bans, and better information to the public on the benefits of a smoke-free society. MPOWER means to monitor tobacco use, protect people from tobacco smoke, offer help to quit tobacco use, warn about the dangers of tobacco, enforce bans on tobacco advertising as well as promotion, and to raise taxes on tobacco products.
Early detection of lung cancerPatients with early-stage lung cancer have better prognosis than those with more advanced disease. Therefore, early detection of lung cancer should improve cure rates and survival of patients. Screening with low-dose CT was recently shown in two large randomized trials to reduce mortality among smokers or former smokers at high risk for lung cancer.5,6 In the National Lung Screening Trial (NLST), lung cancer mortality was reduced by 20% and overall mortality by 6.7% by low-dose CT compared to chest radiographs.5 Three annual screenings were performed in this trial. In the NELSON trial, lung cancer mortality was reduced by 26%.6 Based on these results and those from smaller European trials, lung cancer screening is now endorsed by several scientific societies including the European Society of Radiology and the European Respiratory Society.7
The implementation of early detection and screening by low-dose CT is currently ongoing in several countries and cancer centers with appropriate infrastructure, multidisciplinary expertise and quality control. A multidisciplinary expert panel should assure guidance, monitoring and quality control. The screening population should consist of current or former smokers in accordance with the inclusion criteria of the two randomized trials, although validated risk stratification approaches might play a role in the future. Persons to be screened must be informed about potential benefits and harms of screening, the risk of false-positive and false-negative results, and on the fact that screening is no guarantee for avoiding death from lung cancer. CT examinations including volumetric measurements for assessment of pulmonary nodules must be standardized. Clear definition of positive findings, establishment of algorithms for management of positive or suspicious findings, and establishment of registers for anonymous monitoring of persons are other requirements. Screening programs should also offer smoking cessation advice for active smokers. Early detection and screening programs will most likely reduce mortality rates of lung cancer in the future.
Adjuvant therapy of resected non-small cell lung cancerAdjuvant chemotherapy with cisplatin-based regimens has been re-evaluated within phase 3 trials since 1995 when a meta-analysis indicated a trend towards improved survival for these regimens compared to observation alone.8 Three out of five phase 3 trials demonstrated a survival benefit for cisplatin-based chemotherapy (for review see Ref.9). Among the positive trials, the 5-year survival rates increase by 4–15%.10–13 The Lung Adjuvant Cisplatin Evaluation meta-analysis, which included patients from all five phase 3 trials, confirmed a survival gain at five years of 5.4% for adjuvant cisplatin-based regimens and 8.9% for cisplatin plus vinorelbine.14,15 Therefore, adjuvant chemotherapy with a cisplatin-based doublet, preferentially cisplatin plus vinorelbine, has been established as standard for patients with completely resected tumors and pathological tumor stages 2 or 3.
Strategies to improve outcome of adjuvant therapy focused on the characterization of predictive biomarkers, targeted therapies and tumor vaccines. Predictive biomarkers and customized chemotherapy based on biomarkers remain experimental.16–20 Bevacizumab added to adjuvant chemotherapy failed to increase survival.21 EGFR tyrosine kinase inhibitors (TKIs) also failed to improve survival of patients unselected for EGFR mutations.22,23 However, adjuvant therapy with gefitinib increased disease-free survival compared to chemotherapy in a Chinese study among patients with resected EGFR mutation-positive NSCLC and may be an option for these patients.24 Further trials on adjuvant therapy with EGFR TKIs or ALK inhibitors are ongoing in patients who harbor EGFR mutations or ALK fusions in their cancers. Vaccination with the MAGE-A3 vaccine failed to improve outcome in MAGE-A3-positive patients and resected stage IB-IIIA NSCLC.25 Immune checkpoint inhibitors hold great promise because of their efficacy in metastatic and locally advanced NSCLC and are currently evaluated within phase 3 trials in patients with completely resected NSCLC and tumor stage IB (<4 cm) – IIIA (for review see Ref.9). Within these trials, patients receive adjuvant chemotherapy followed by an immune checkpoint inhibitor as single agent for one year. Primary endpoints of the trials are often disease-free survival. Finally, surrogate endpoints would be of interest in order to shorten the duration of adjuvant trials. Residual disease based on circulating tumor DNA at the end of adjuvant chemotherapy could be such an endpoint and should be further studied.
Induction chemotherapy of operable NSCLCInduction chemotherapy with a platinum-based doublet prior to surgery resulted in survival benefits similar to the ones achieved with adjuvant chemotherapy in patients with operable NSCLC.26 Therefore, induction chemotherapy is a valid treatment option for patients with operable NSCLC, particularly for those with marginally resectable tumors. Current clinical trials evaluate tyrosine kinase inhibitors as induction therapy among patients with driver mutation-positive NSCLC. Immune checkpoint inhibitors are also evaluated as induction therapy, either alone or in combination with chemotherapies.
Treatment of locally advanced NSCLCPatients with locally advanced NSCLC require both local and systemic treatments and, therefore, multidisciplinary co-operation is crucial for their optimal care.27
Patients with completely resected tumors receive adjuvant chemotherapy. Selected patients, particularly those with marginally resectable tumors, are candidates for induction chemotherapy followed by local treatment. For the majority of patients, however, chemoradiotherapy remains standard treatment.27,28 Concomitant chemoradiotherapy is superior over the sequential approach.29 Consolidation therapy with durvalumab has recently been approved for patients with response or stable disease after chemoradiotherapy and PD-L1 levels ≥1% in their tumors. This approval was based on results of the PACIFIC trial which demonstrated improved disease-free and overall survival for consolidation therapy with durvalumab.30 High dose conformal radiotherapy and the addition of cetuximab to chemoradiotherapy failed to improve outcome of patients.31
Two major therapeutic strategies to improve outcome are currently studied within clinical trials. The first strategy focuses on the integration of immune checkpoint inhibitors. These drugs are evaluated as induction therapy, either as single agent or combined with induction chemotherapy, and also in combination with radiotherapy or chemoradiotherapy. Similarly, EGFR tyrosine kinase inhibitors are evaluated as induction therapy in patients with EGFR mutation-positive NSCLC. There is great hope that these strategies will improve survival of patients with locally advanced NSCLC in the future, although there is also concern that some of these combined treatments might result in unacceptable toxicity.
Treatment of advanced NSCLCPatients with advanced NSCLC receive palliative therapies with systemic treatments and, in case of local problems, radiotherapy or surgery. Systemic anticancer treatments are chemotherapy, targeted therapies and immune checkpoint inhibitors. The type of systemic therapy depends on tumor histology, presence or absence of driver mutations in tumors, performance status of patients and other factors. Supportive care including end-of-life care also plays a major role in patients with advanced NSCLC.
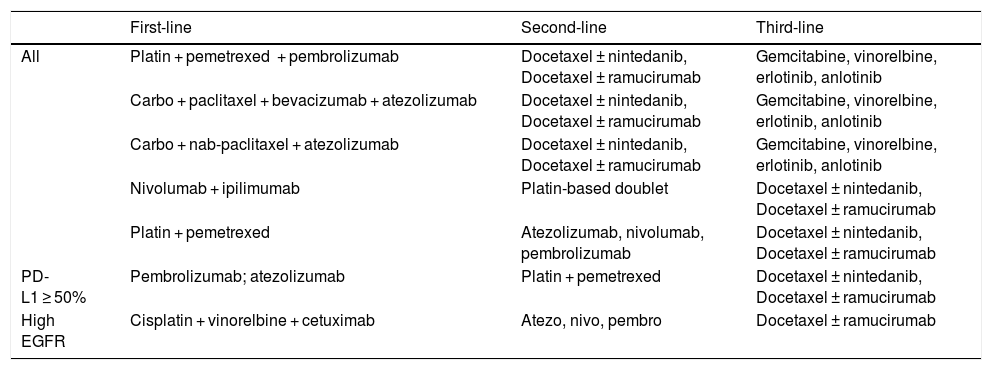
Advanced driver mutation-negative NSCLCPatients with advanced NSCLC have received first-line chemotherapy, maintenance chemotherapy and second-line therapy for many years.32–34 Immune checkpoint inhibitors have recently become part of the standard treatment for patients with advanced driver mutation-negative NSCLC.35 They were initially approved as single agents for patients who had been pretreated with chemotherapy. Then immune checkpoint inhibitors became established in the first-line setting, either as single agents or in combination with chemotherapy. Current treatment options for patients with non-squamous and squamous NSCLC are shown in Tables 1 and 2.
Treatment of advanced driver-negative non-squamous NSCLC.
| First-line | Second-line | Third-line | |
|---|---|---|---|
| All | Platin + pemetrexed + pembrolizumab | Docetaxel ± nintedanib, Docetaxel ± ramucirumab | Gemcitabine, vinorelbine, erlotinib, anlotinib |
| Carbo + paclitaxel + bevacizumab + atezolizumab | Docetaxel ± nintedanib, Docetaxel ± ramucirumab | Gemcitabine, vinorelbine, erlotinib, anlotinib | |
| Carbo + nab-paclitaxel + atezolizumab | Docetaxel ± nintedanib, Docetaxel ± ramucirumab | Gemcitabine, vinorelbine, erlotinib, anlotinib | |
| Nivolumab + ipilimumab | Platin-based doublet | Docetaxel ± nintedanib, Docetaxel ± ramucirumab | |
| Platin + pemetrexed | Atezolizumab, nivolumab, pembrolizumab | Docetaxel ± nintedanib, Docetaxel ± ramucirumab | |
| PD-L1 ≥ 50% | Pembrolizumab; atezolizumab | Platin + pemetrexed | Docetaxel ± nintedanib, Docetaxel ± ramucirumab |
| High EGFR | Cisplatin + vinorelbine + cetuximab | Atezo, nivo, pembro | Docetaxel ± ramucirumab |
Treatment of advanced driver-negative squamous NSCLC.
| First-line | Second-line | Third-line | |
|---|---|---|---|
| All | Platin-based CT + pembrolizumab | Docetaxel ± ramu, afatinib | Afatinib, gemcitabine, vinorelbine, anlotinib |
| Carbo + nab-pacl + atezolizumab | Docetaxel ± ramucirumab | Afatinib, erlotinib, gem, vinorelbine, anlotinib | |
| Nivolumab + ipilimumab | Platin-based doublet | Docetaxel ± nintedanib, Docetaxel ± ramucirumab | |
| PD-L1 ≥ 50% | Pembrolizumab; atezolizumab | Platin-based doublet | Docetaxel ± ramucirumab |
| All | Platin-based CT | Atezo, nivo, pembro | Docetaxel ± ramucirumab |
| High EGFR | Platin + gemcitabine + necitumumab | Atezo, nivo, pembro | Docetaxel ± ramucirumab |
Platinum-based doublets have been standard first-line chemotherapy for patients with advanced NSCLC for many years.32,33 These doublets include one of the following third-generation cytotoxic drugs: vinorelbine, gemcitabine, pemetrexed, paclitaxel, nab-paclitaxel and docetaxel. First-line platinum-based chemotherapy relieves cancer-related symptoms and increases median survival by 1.5 months and the 1-year survival rate by 9%.36
Cisplatin-based doublets are slightly superior to carboplatin regimens37 and are preferred for patients with good performance status. Carboplatin-based doublets are preferred in elderly patients, patients with impaired organ (kidney, heart) functions or whenever ease of administration is of particular importance. First-line chemotherapy is combined with bevacizumab in selected patients with non-squamous NSCLC.38 Chemotherapy combined with cetuximab or necitumumab improved survival in patients with NSCLC and squamous cell NSCLC, respectively.39,40 Patients with high EGFR expression or EGFR FISH-positivity of tumors particularly benefited from the addition of EGFR antibodies to chemotherapy.41–43 Elderly patients and patients with reduced performance status are treated with single agents or well tolerated doublets.44
The establishment of immune checkpoint inhibitors has recently changed the therapeutic landscape in patients with advanced NSCLC.35 Immune checkpoint inhibitors were studied in the first-line setting as single agents and in combination with chemotherapy. Pembrolizumab or atezolizumab improved overall survival compared to platinum-based doublets among patients with PD-L1 expression in ≥50% of tumor cells.45,46 First-line chemotherapy combined with either pembrolizumab or atezolizumab improved progression-free survival and, in some studies, also overall survival compared to chemotherapy alone.47–52 Although the benefit from immune checkpoint inhibitors appeared to increase with increasing PD-L1 expression of tumor cells, patients with PD-L1 expression in <1% of tumor cells also derived clinically meaningful improvements from the addition of immune checkpoint inhibitor to platinum-based doublets.53 Based on these results, chemoimmunotherapy replaced chemotherapy as standard first-line therapy in patients with advanced driver mutation-negative NSCLC.35 Patients with good performance status now receive a platinum-based doublet plus an immune checkpoint inhibitor regardless of PD-L1 levels of tumors (Tables 1–2). Strategies to improve clinical outcome of patients focus on novel drugs which may further enhance the immune response towards tumors. These drugs are studied as single agents or in combination with current standard treatments.
Maintenance therapy and treatment at the time of disease progressionMaintenance therapy with pemetrexed is established as a valid treatment option for selected patients with non-squamous cell NSCLC. Bevacizumab, necitumumab and immune checkpoint inhibitors are usually continued as maintenance or consolidation therapy after completion of first-line chemotherapy.
Patients who progress after platinum-based chemotherapy are treated with docetaxel plus/minus ramucirumab, docetaxel plus/minus nintedanib, pemetrexed, erlotinib or afatinib.33,54–56 Those patients who had not been treated with an immune checkpoint inhibitor in the first-line setting should receive one of them as second-line therapy.
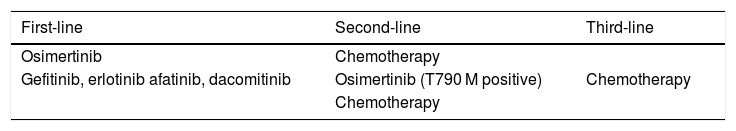
Advanced driver mutation-positive NSCLCThe characterization of driver mutations and the subsequent establishment of corresponding TKIs as standard first-line treatment for patients with advanced driver mutation-positive NSCLC have been milestones in the treatment of patients with lung cancer. EGFR mutations, ALK translocations, ROS1 aberrations and BRAF mutations are currently routinely assessed in advanced NSCLC, particularly in adenocarcinomas. Other molecular aberrations are assessed dependent on availability of tests and corresponding drugs. While tumor tissue is currently the main source for molecular analyses, liquid biopsies will gain importance for diagnosis and particularly disease monitoring in the future.57
EGFR TKIs have established themselves as standard first-line treatment for patients with advanced EGFR mutation-positive NSCLC (Table 3). First- and second-generation EGFR TKIs resulted in superior progression-free survival compared to chemotherapy among patients with advanced EGFR mutation-positive NSCLC (for review see Refs.58–60). Osimertinib, a third-generation TKI, improved progression-free and overall survival compared to gefitinib or erlotinib in previously untreated patients61 and, therefore, has become the preferred first-line therapy.
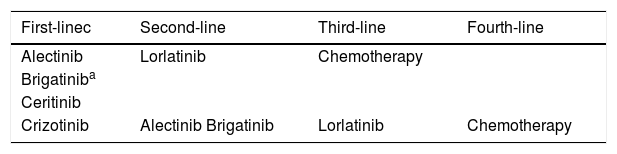
Several ALK inhibitors have also been established for patients with advanced ALK-positive NSCLC (for review see Refs.62,63). They include crizotinib, alectinib, ceritinib, brigatinib and lorlatinib. Crizotinib was the first ALK inhibitor to be approved.64,65 Second-generation ALK inhibitors have broader efficacy as well as better penetration into the brain and have become the preferred first-line therapy.66–68 Alectinib and brigatinib resulted in longer progression-free survival compared to crizotinib in the first-line setting.66,67 The third generation inhibitor lorlatinib has shown efficacy in treatment-naive patients and patients who have developed resistance to crizotinib or second-generation ALK inhibitors.69 Therefore, lorlatinib has recently been approved for patients whose disease has progressed after alectinib or ceritinib, or after crizotinib plus at least another ALK inhibitor. A proposal for treatment of ALK-positive patients is shown in Table 4. In routine clinical practice, the selection of an ALK inhibitor should be based on its availability as well as re-imbursement, presence of brain metastases, doctor’s judgement and patient preference. The optimal sequencing of ALK inhibitors, however, has yet to be determined within clinical trials.
Treatment of advanced ALK-positive NSCLC.
| First-linec | Second-line | Third-line | Fourth-line |
|---|---|---|---|
| Alectinib | Lorlatinib | Chemotherapy | |
| Brigatiniba | |||
| Ceritinib | |||
| Crizotinib | Alectinib Brigatinib | Lorlatinib | Chemotherapy |
Patients with extensive stage SCLC are now treated with platinum plus etoposide in combination with an immune checkpoint inhibitor. This change from chemotherapy to chemoimmuntherapy is based on results from two phase 3 trials which demonstrated increased overall survival for chemotherapy plus atezolizumab or durvalumab compared to chemotherapy alone among patients with extensive stage SCLC.70,71 Patients with limited stage SCLC continue to receive first-line therapy with cisplatin plus etoposide and thoracic radiotherapy. Patients should also be considered for prophylactic cranial irradiation. At the time of disease progression, topotecan is established as standard therapy.