The assessment of inspiratory effort in the patient with acute hypoxaemic respiratory failure (AHRF) can be performed invasively by measuring the oesophageal pressure swing (ΔPes) during the inspiratory act.1 The reduction in ΔPes after two hours of non-invasive ventilation (NIV) in patients with AHRF is a predictor of NIV success.2
The study of diaphragm thickening fraction (TFdi) is a predictor of NIV failure in patients hospitalised for COPD exacerbation with respiratory acidosis.3
A correlation between TFdi and ΔPes has been demonstrated in patients undergoing different levels of support during invasive (IMV)4 and NIV.5 So far, the validity of TFdi vs. ΔPes in recording the patient's effort during Continuous airways pressure (CPAP) and High Flow Nasal Cannula (HFNC), where any form of inspiratory support is lacking, has not yet been assessed.
De Vita et al. demonstrated that the values of PaO2, SpO2, respiratory rate, recorded during Venturi mask oxygen therapy and during CPAP are associated with CPAP failure.6 However, finding predictors of failure of non-invasive respiratory supports (NRS) has always been a challenge. For this reason, between February and May 2021, we conducted a randomized short-term physiological study within the first 24 h of hospital admission to compare the effects of standard oxygen therapy, HFNC, CPAP and NIV on breathing pattern, gas exchange, inspiratory effort, and dynamic transpulmonary pressure (PLDyn) in patients with moderate-to-severe AHRF due to COVID-19 pneumonia.7
This research letter is a new analysis of our previous investigation in which we compare the inspiratory effort during standard oxygen therapy (SOT) and 3 forms of NRS, using both the Oesophageal pressure swing (ΔPes) and the diaphragm ultrasound.
The local Ethic Committee approved the study (691/2020/Sper/AOUBo) and written informed consent was obtained from all the patients. The study was registered at the Clinical Trial Registry (NCT04741659).
We considered eligible any adult patient (≥ 18 years old) with AHRF and a PaO2/FiO2 ratio < 200 mmHg evaluated during spontaneous unassisted breathing trial (FiO2 of at least 0.40), due to pneumonia and a confirmed molecular diagnosis of COVID-19. Exclusion criteria were: previous clinical, radiological and histological diagnosis of pneumopathy, body mass index > 30 kg/m2; known diagnosis of sleep-disorders, restrictive pulmonary/chest wall disease, cardiac arrest, severe haemodynamic instability (> 1 vasoactive amine for at least 24 h), acute coronary syndrome (unstable angina/IMA), severe arrhythmias, inability to protect the airway, respiratory arrest and need for intubation, pregnancy or suspected, use of sedative drugs, long-term home oxygen therapy.
Patients underwent trials of 30 min each where they were randomly alternated with CPAP, HFNC and NIV; between each trial, there was a 30-min wash-out period where SOT was administered, as described in ref.6 TFdi was measured using a linear probe at functional residual capacity and after each tidal inspiratory act. TFdi was calculated as follows:
After excluding the presence of right or left chronic elevated hemidiaphragm and/or paralysis by ultrasound, at each protocol step, two investigators performed bedside ultrasound evaluation of the right hemidiaphragm on the zone of apposition between the anterior and medial axillary lines,8 with the patient in semi recumbent position. Each investigator performed and recorded three sets of measurements independently, which were then averaged.
Measurements of oesophageal pressure (Pes) were obtained by placing a nasogastric tube equipped with an oesophageal balloon in the lower 1/3 of the oesophagus. Oesophageal pressure swing was calculated as the maximum decrease or increase in Pes from the end-expiratory level. The study of patient's tidal volume and minute ventilation (Vt and Ve) was obtained by respiratory inductance plethysmography. Measurements were obtained during each trial and during SOT.
Relationships between variables TFdi, ΔPes and Ve were studied using the Spearman correlation. Differences in TFdi values between groups were analyzed by one-way analysis of variance (ANOVA). P values lower than 0,05 were considered statistically significant. The IBM SPSS Statistics software (version 21) was used for data analysis.
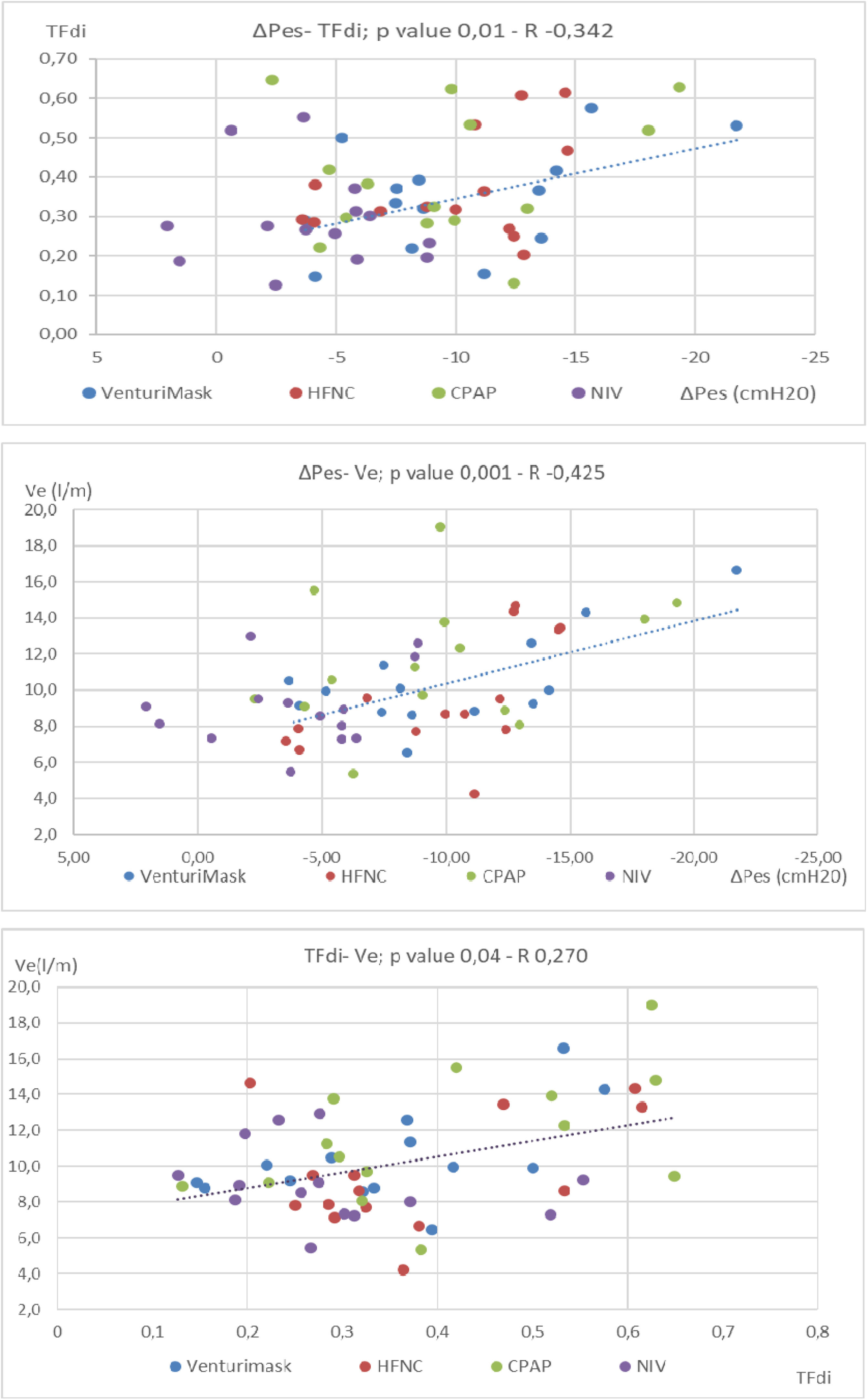
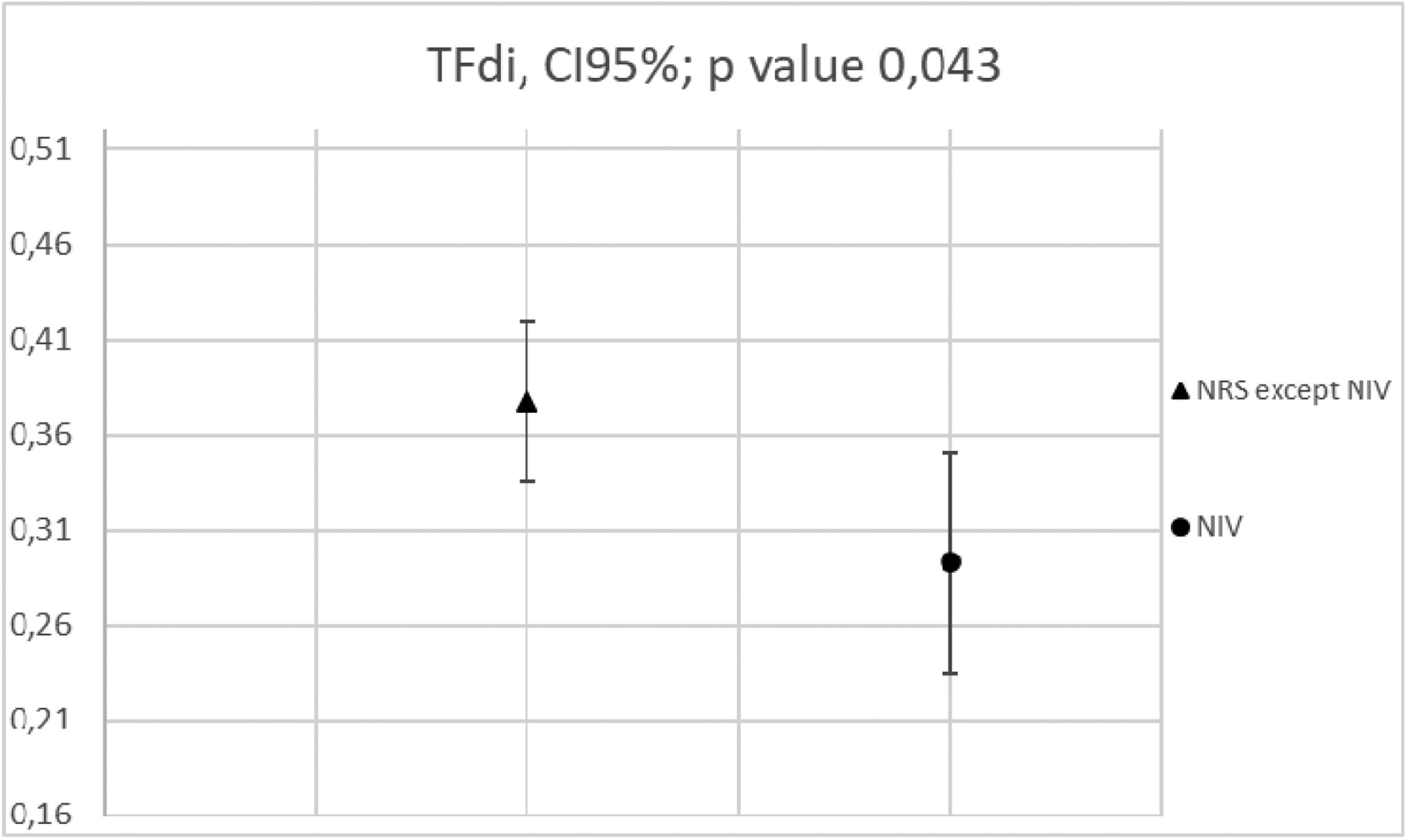
Fifteen patients were enrolled. Patient's mean age was 63 ±9 years old, mean SAPS II and APACHE scores at admission were 22±6 and 8,7±2, respectively. Mean PaO2/Fio2 ratio was 126±34 and mean respiratory rate was 24.6±5.4 breaths per minute.7 There was a direct correlation between ΔPes and TFdi detected by ultrasound, regardless of the type of NRS used (Fig. 1 upper part; p value 0,01 R = -0,342). The Ve was found to be directly correlated to the patient's TFdi (p value 0,04 R = 0,270) and ΔPes (p value 0,001 R = -0,425) (Fig. 1, middle and lower part). The use of NIV resulted in a significant reduction in TFdi developed by each patient compared to all other NRS used (respectively mean TFdi = 0,292±0,12 versus 0,377 ±0,142 p value 0,043, Fig. 2).
Upper part: Spearman Correlation analysis between TFdi and ΔPes (p value 0,01): inspiratory effort measured by oesophageal manometry was directly correlated to TFdi, regardless of the type of NRS applied. Each dot corresponds to the mean value of 6 measurements of ΔPes and their linear correlation with TFdi, for each patient during each different NIRS trial (colors).
Middle part: Spearman correlation analysis between Ve and ΔPes (p value 0,001). Each dot corresponds to the mean value of 6 measurements of ΔPes and their linear correlation with VE, for each patient during each different NIRS trial (colors).
Lower Part: Spearman correlation analysis between VE and TFdi (p value 0,04). Each mark corresponds to the mean value of 6 measurements of TFdi and their linear correlation with VE, for each patient during each different NIRS trial (colors).
ΔPes: oesophageal pressure swing; TFdi: diaphragm thickening fraction; VE: minute ventilation; HFNC; High flow nasal cannula, CPAP; continuous positive airway pressure, NIV; non-invasive ventilation.
Lower part: correlation analysis between Ve and ΔPes (p value 0.001) and TFdi (p value 0,04).
Each mark corresponds to the mean value of 6 measurements of ΔPes and TFdi (triangles and dots, respectively) and their linear correlation with Ve, for each patient during each different NIRS trial (colors).
ΔPes: oesophageal pressure swing; TFdi: diaphragm thickening fraction; Ve: minute ventilation; HFNC; High flow nasal cannula, CPAP; continuous positive airway pressure, NIV; non-invasive ventilation.
Effects of Pressure support application on TFdi (one-way ANOVA). NIV was the only NRS to significantly reduce TFdi compared to VenturiMask, HFNC, or CPAP (p value 0,043). TFdi: diaphragm thickening fraction; NIV; non-invasive ventilation; NRS: non invasive respiratory support. HFNC= high flow nasal cannula; CPAP= continuous positive airway pressure.
To the best of our knowledge, this is the first study comparing the extent of inspiratory effort assessed by ΔPes and TFdi of the diaphragm during the 3 major forms of NRS.
In patients with AHRF receiving NIV, the evaluation of patients’ inspiratory effort is desirable to predict NIV failure and to avoid a delay in endotracheal intubation.2 Nonetheless, assessing ΔPes is not always feasible since it is not widely used in clinical practice. Alternatively, ultrasound study of TFdi is a simple, reproducible method,9 which can be performed at the patient's bedside. The correlation analysis in our first study7 revealed that VE was directly correlated with the patient's inspiratory effort (p value 0,001): patients with higher ΔPes experienced an increase in VE. In line with these findings, here an increase in VE was directly proportional to the TFdi.
Our results showed that there was a moderate correlation between ΔPes and TFdi. This can be explained by the fact that the change in ΔPes is influenced not only by diaphragmatic contraction, but also by the contraction of all the inspiratory muscles. The measurement of transdiaphragmatic pressure (Pdi) may have a better correlation with TFdi. Recent studies, however, show controversy even when comparing these two variables.10,11 For instance, Puolard et al. found that the Pdi was weakly related to diaphragm thickening fraction.10 Furthermore, even when diaphragmatic contractility is equal, oesophageal pressure changes may be influenced by a different ratio of chest wall and lung elastance.
In the absence of an esophageal balloon to monitor ΔPes, this modest correlation between ΔPes and TFdi would allow us to determine relative changes in the patient's inspiratory effort over time and following the application of different types of NRS: NIV was the only support that significantly reduced ΔPes.7 Similarly, the use of a pressure support (NIV) resulted in a reduction of the TFdi, when compared to any other NRS.
This research has limitations. First, it represents a physiological study with a small sample size. Second, diaphragm sonographic assessment could be influenced by the operator. However, to overcome this limitation, ultrasound assessment was performed independently by two operators, and, consistent with previous studies,12 the interclass correlation was found to be high, reducing the risk of bias. Third, as we mention before, we have not correlated TFdi with the Pdi, since we did not measure it.
In absence of diaphragm dysfunction,13 TFdi can be used to estimate a change in patients’ inspiratory effort during NRS, similar to what Umbrello demonstrated in patients undergoing IMV.4 Further studies are needed to assess the correlation between the TFdi and the patients’ clinical outcomes.
The authors thank Gilda Giancotti for technical assistance.