Transbronchial lung cryobiopsy (TBLC) is frequently described for the diagnosis of diffuse parenchymal lung diseases (DPLD). A few studies have reported transbronchial cryobiopsy for the diagnosis of peripheral pulmonary lesions (PPL...s). We aimed to study the utility and safety of transbronchial cryobiopsy for the diagnosis of PPL...s.
MethodsWe performed a systematic search of the PubMed and Embase databases to extract the relevant studies. We then performed a meta-analysis to calculate the diagnostic yields of transbronchial cryobiopsy and bronchoscopic forceps biopsy.
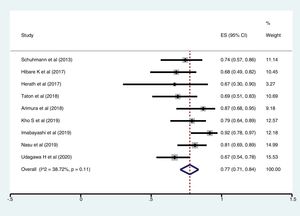
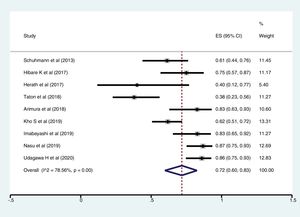
ResultsFollowing a systematic search, we identified nine relevant studies (300 patients undergoing cryobiopsy). All used Radial Endobronchial Ultrasound (R-EBUS) for PPL localization. The pooled diagnostic yield of transbronchial cryobiopsy was 77% (95% CI, 71%...84%) (I^2=38.72%, p=0.11). The diagnostic yield of forceps biopsy was 72% (95% CI, 60%...83%) (I^2=78.56%, p<0.01). The diagnostic yield of cryobiopsy and forceps biopsy was similar (RR 1.05, 95% CI 0.96...1.15), with a 5% risk difference for diagnostic yield (95% CI, ...6% to 15%). There was significant heterogeneity (I^2=57.2%, p=0.017), and no significant publication bias. One severe bleeding and three pneumothoraxes requiring intercostal drain (ICD) placement (major complication rate 4/122, 1.8%) were reported with transbronchial cryobiopsy.
ConclusionsR-EBUS guided transbronchial cryobiopsy is a safe and efficacious modality. The diagnostic yields of TBLC and forceps biopsy are similar. More extensive multicentre randomized trials are required for the further evaluation and standardization of transbronchial cryobiopsy for PPL...s.
Peripheral pulmonary lesions (PPL...s) are a frequent indication for pulmonologist referral in clinical practice. Clinicians can incidentally find PPL...s on thoracic imaging, and many are detected on CT scans performed for lung cancer screening. The diagnosis is challenging especially for small lesions and those located in proximity to major vascular structures.1 An accurate diagnosis is essential so that treatment decisions can be optimized. Transthoracic percutaneous biopsy and bronchoscopy are the primary modalities for tissue biopsy of PPL...s. The decision regarding the choice of approach for biopsy depends on the proximity of the lesion to the chest wall, availability of resources, risk of complications depending on the patient...s clinical status and physician preference. Percutaneous transthoracic approaches (CT or ultrasound-guided biopsy) provide a superior diagnostic yield.2 However, the risk of pneumothorax is higher, especially in patients with emphysema.
Bronchoscopic modalities are safer with a lower risk of pneumothorax. Endobronchial anatomy can also be evaluated. Conventional bronchoscopy for PPL...s provides a low diagnostic yield.3 Radial Endobronchial Ultrasound (R-EBUS) is a useful modality for guided bronchoscopic biopsy of PPL...s. However, the material obtained with conventional forceps biopsy under R-EBUS guidance is often insufficient.4 The overall diagnostic yield of R-EBUS for PPL...s (70.6%) is not excellent and requires further improvement.5 Various innovations have attempted to optimize the yield of bronchoscopic biopsy of PPL...s. These technologies include guide sheath method, bronchoscopic navigation, fluoroscopy, cone-beam CT guidance, and robotic bronchoscopy.6 Although the newer technologies allow a more accurate guide to the target lesion, a suboptimal diagnostic yield compared with CT guided percutaneous biopsy remains a significant limitation.
Transbronchial lung cryobiopsy (TBLC) is commonly used in the evaluation of diffuse parenchymal lung disease (DPLD)...s.7,8 The samples obtained with TBLC are significantly larger than the conventional bronchoscopic forceps biopsy. A few studies have described the utility of R-EBUS guided transbronchial cryobiopsy for the evaluation of PPL...s. Some have also compared the yield of cryobiopsy with conventional forceps biopsy. There is increasing interest in the use of bronchoscopic cryobiopsy for sampling PPL...s. The advantage of the cryoprobe is the ability to obtain a more extensive tissue, and a 360../lateral biopsy.9 For adjacent or eccentric lesions, this ability may be particularly advantageous. Theoretically, the freezing of a larger area surrounding the tip of the cryoprobe may also be beneficial in smaller nodules. The risks with TBLC for DPLD...s include pneumothorax and bleeding. In contrast to TBLC for diffuse interstitial lung diseases, the technique of cryobiopsy for PPL...s has not been standardized.10
We carried out a systematic review of the literature to identify the studies describing the yield and safety of transbronchial cryobiopsy for PPL...s. We included studies which included human subjects undergoing transbronchial cryobiopsy for PPL. Both prospective and retrospective studies were eligible for inclusion. We also performed a meta-analysis to calculate the pooled diagnostic yield of cryobiopsy and its comparison with forceps biopsy. We also summarise the complications and safety of bronchoscopic cryobiopsy for PPL...s.
MethodsSearch strategy and initial reviewTwo authors (K.M and S.PB) performed a systematic search of the two databases: PubMed and EMBASE (01.01.2004 up to 12.08.2020) to identify the original, peer-reviewed, full-length, human subject articles describing the use of transbronchial cryobiopsy for PPL...s. We used the following database-specific search strategy. Free text search terms were: (cryoprobe OR cryobiopsy OR cryotherapy OR cryo OR tblc OR transbronchial lung cryobiopsy) AND (peripheral pulmonary lesion OR ppl OR solitary pulmonary nodule OR lung nodule OR pulmonary nodule OR radial ebus OR ebus OR radial endobronchial ultrasound OR endobronchial ultrasound OR guide sheath OR radial probe OR virtual bronchoscopic navigation OR vbn OR emn OR electromagnetic navigation). All the retrieved studies were imported into reference management software (EndNote). Duplicate citations were discarded. The studies were screened by title and abstract. Full texts were downloaded for review, wherever required. The reference lists of the extracted studies were also reviewed. The finally selected studies were screened independently by three authors (K.M, S.M and S.PB). Studies describing the utilization of cryobiopsy for peripheral pulmonary lesions were included. For inclusion, sufficient data for calculating the diagnostic yield of cryobiopsy for PPL were required. The following studies were excluded: (a) studies that did not report the utilization of cryobiopsy for PPL (b) studies not in English language (c) review articles, editorials, abstracts, and letters without any case description (d) case reports or series with fewer than five patients. Any disagreement between the authors was resolved after mutual discussion.
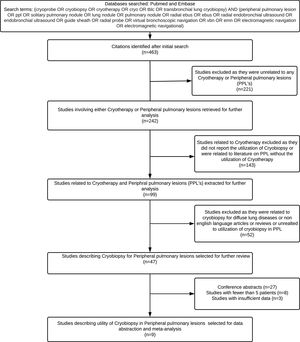
Data abstractionData from the finally selected studies were abstracted on a data extraction form. The following information was retrieved after a thorough review of the full text ..÷ (a) author, (b) year, (c) number of patients, (d) gender, (e) number of centres, (f) study design, (g) age, (h) type of cryoprobe, bronchoscope and forceps used (i) anaesthesia, (j) size of target lesion (k) use of navigation, (l) use of fluoroscopy, (m) number of biopsies (n) location of the target lesion, (o) diagnostic yield of cryobiopsy, (p) diagnostic yield of forceps biopsy, (q) complications. The systematic review methodology is summarized in Fig. 1. Complications were considered minor if they included mild to moderate bleeding responding to local measures as reported by the author. Major complications included pneumothorax and severe bleeding, as described in the original study.
Assessment of study qualityStudy quality was evaluated with the Methodological Index for Non-Randomized Studies (MINORS) and the Cochrane tool for Randomized studies. The MINORS tool consists of a checklist of eight items developed for non-comparative studies and four additional items for comparative studies. Items on the MINORS tool are scored as 0 (not reported), 1 (reported but inadequate) and 2 (reported and adequate), resulting in a total score of 16 for non-comparative studies and 24 for comparative studies.11 The Cochrane tool for RCT consists of seven questions and bias for each item is rated as ..úLow risk..Ñ, ..úHigh risk..Ñ, or ..úUnclear risk..Ñ to judge.12 Each study was independently reviewed by two authors (KM and SPB), after which the scores were compared and decided on final scores during a consensus meeting.
Statistical analysisStatistical analyses were performed using the STATA statistical analysis software (StataCorp 2017. Stata Statistical Software: Release 15. College Station, TX: StataCorp LLC) and RevMan (Review Manager [Computer program]. Version 5.3. Copenhagen: The Nordic Cochrane Centre, The Cochrane Collaboration, 2014). A proportional meta-analysis was performed to calculate the pooled diagnostic yield of forceps biopsy and transbronchial cryobiopsy. Pooled proportions, along with 95% confidence intervals were derived. A meta-analysis was also performed to compare the diagnostic yield of forceps biopsy and cryobiopsy using the pooled RR (relative risk) method. The pooled risk difference (RD) of the yield of cryobiopsy and forceps biopsy was also calculated. Complications were classified into a minor, or a major complication and summary statistics were reported.
Heterogeneity assessmentThe impact of heterogeneity on the pooled estimates of the diagnostic yields and the comparative diagnostic yield was assessed using the I2 (I-Square) test Cochran Q statistic.13 I2 is a statistical tool to evaluate the impact of unobserved heterogeneity. It describes the percentage of total variation seen across studies that are attributable to heterogeneity rather than chance. An I2 value of ...50% indicates significant heterogeneity. For the Cochran Q statistic, a p-value <0.1 is significant for the presence of statistical heterogeneity.
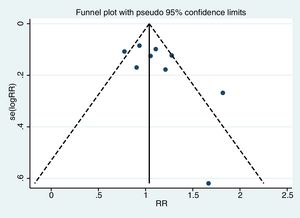
Assessment of publication biasThe funnel plot (Egger test) was used to assess for publication bias (statistically significant publication bias when p<0.1).14
ResultsThe initial literature search yielded 124 articles from which nine studies (457 patients, 300 underwent cryobiopsy) were selected for data abstraction and included in the meta-analysis. The baseline study characteristics are summarized in Tables 1 and 2. The methodological quality of the studies is summarized in Table 3. Of the nine studies, four were retrospective,9,15...17 and five were prospective studies (including one randomized controlled trial, RCT).4,18...21 All were single-centre studies. Most studies reported a forceps biopsy group as a comparator in the same group of patients. One study reported the yield of forceps biopsy performed by the same operator in a different set of patients.9
Baseline characteristics of studies describing the utility of transbronchial cryobiopsy for PPLs.
| No. | Author/year | Country | Study design | Total number (number undergoing cryobiopsy) | Inclusion criteria | Comparison with forceps biopsy with cryobiopsy in same patients | Age (years) | Size of PPL (mm) | % Of lesions in upper lobes |
|---|---|---|---|---|---|---|---|---|---|
| 1 | Schuhmann et al./20144 | Germany | RCT | 38 (31) | Solid pulmonary lesion of <40mm | Yes | Median IQR, 68 (37...84) | 29.7 (7.3) | NA |
| 2 | Hibare et al./201715 | India | Retrospective | 55(28) | PPL requiring R-EBUS, bronchoscopically invisible | Yes | Mean (SD), 61.8 (7.2) (M), 59.3 (11.2) (F) | Size data for 41 (28>3cm, 13<3cm) | NA |
| 3 | Herath et al./201819 | New Zealand | Prospective | 6 (6) | PPL 1cm or above on CT | Yes | Mean (SD), 56.66 (13.14) | 41 (19...66) | 66.7 |
| 4 | Taton et al./201820 | Belgium | Prospective | 32 (29) | Age over 18 years; a CT-detected solid or nonsolid nodule with a diameter from 8 to 20mm | Yes | Mean (SD), 68(9) | 16 (3) | 51.7 |
| 5 | Arimura et al./201918 | Japan | Prospective | 23 (23) | Solid lesions >2cm away from pleura, bronchoscopically invisible | Yes | Median IQR 69.5 (46...82) | 36 (10...81) | 60.9 |
| 6 | Kho et al./20199 | Malaysia | Retrospective | 114 (38) | All adult patients undergoing R-EBUS-guided transbronchial biopsy | No | Median IQR 56 (47.8...64.5) | 34.8 (26.3...45.1) | 23 (60.5) in cryobiopsy group46 (60.5) in the forceps group |
| 7 | Imabayashi et al./201916 | Japan | Retrospective | 38 (36) | Suspected peripheral lung cancer undergoing cryobiopsy | Yes | Median IQR 66.9 (44...81) | 37.2..19.4 | 52.8 |
| 8 | Nasu et al./201917 | Japan | Retrospective | 53 (53) | A final diagnosis of lung cancer in PPL who underwent cryobiopsy | Yes | Median IQR 75 (41...90) | 32 (8...85) | 50.9 |
| 9 | Udagawa et al./202021 | Japan | Prospective | 121 (57) | Aged 20...80 years with suspected or diagnosed primary lung cancer by chest computed tomography scheduled to undergo TBB by bronchoscopy | Yes | Median IQR 68 (31...79) | 38 (Median) | 40.3 |
PPL: peripheral pulmonary lesion; R-EBUS: Radial Endobronchial Ultrasound; TBB: transbronchial biopsy.
Procedural characteristics of studies included for systematic review and meta-analysis.
| No. | Author/year | Modality for sampling | R-EBUS probe used | R-EBUS probe size (mm) | Anaesthesia | Artificial airway | Cryoprobe diameter (mm) | Freezing | No. of cryobiopsies | No. of flexible biopsies | Size of cryobiopsy | Size of forceps biopsy | Comments |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Time (s) | Median (Range) | ||||||||||||
| Mean (SD)/range | |||||||||||||
| 1 | Schuhmann et al./20144 | Radial EBUS+GS | UM-S20-20R; Olympus | 1.7 | GA | Yes, rigid bronchoscopy | 1.2 | 4 | 3 | 3 | 11.17mm2 (1.25...38.59) | 4.69mm2 (0.53...22) | 7 out of 38, PPL not localized in 20min with R-EBUS and were excluded. Duration of the cryobiopsy was significantly longer compared with forceps biopsy. |
| 2 | Hibare et al./201715 | Radial EBUS..GS..Fluoroscopy | UM-S20-17S; Olympus | 1.4 | NA | No | 1.9 | 4 | 3 | 3 | NA | NA | 9 (14%) cases the lesion could not be located by R-EBUS |
| 3 | Herath et al./201719 | Radial EBUS+GS | UM-S20-20R; Olympus | 1.7 | Conscious sedation (5), GA (1) | Yes, GA in one patient, Five not intubated | 1.9 | 4 | 1...3 | NA | 6.4mm | 3.4mm | All the PPLs were visualized as concentric lesions on R- EBUS. To enable contact with the lesion, the GS was trimmed by 3cm from the distal end |
| 4 | Taton et al./201820 | Radial EBUS+GS | UM-S20-17S; Olympus | 1.4 | GA and paralysis (Remifentanil, propofol, rocuronium) | Yes, rigid bronchoscope | 1.9 | 7...8 | 2 | 6 | 5.3..0.7 (mm) | 1.1..0.6 (mm) | The lobar location of the nodule, the bronchus sign, the nodule size, the malignant vs benign disease, or the technique used (nodule visualization or not with EBUS mini probe) for visualizing the nodule in addition to ENB had no statically significant impact on the diagnostic performance |
| 5 | Arimura et al./201918 | Radial EBUS+GS+Fluoroscopy | UM-S20-20R; Olympus | 1.7 | Deep sedation | Yes, intubated 7.5mm ET | 1.9 | 3...5 | 1...2 | 5 | 0.078..0.008 (mean..SEM) cm3 | 0.003..0.0003 (mean..SEM)cm3 | Bronchoscopists performed the procedures with at least three years of experience |
| 6 | Kho et al./20199 | Radial EBUS ..GS..Fluoroscopy | UM- S20-17S, UM-S20-20R, Olympus | 1.4, 1.7 | Conscious sedation or TIVA | Yes (rigid or ETT), at operator...s discretion, 16/38 in cryobiopsy group and 7/76 in forceps group | 1.9 | 3...4 | 5 | NA | NA | ROSE during Cryo with EBUS-GS had a high sensitivity, specificity, PPV, and diagnostic accuracy for PPLs. | |
| 7 | Imabayashi et al./201916 | Radial EBUS without sheath | UM-S20-17S; Olympus | 1.4 | Conscious sedation (Midazolam and Fentanyl) | No | 1.9 | 3.3 (0.7) | 1.5..0.6 (1...3) | NA | 12.2mm2 (5.6) | NA | Stamp cytology with CB facilitates the on-site confirmation of tumor inclusion and improved diagnostic yield. The EBUS probe was located within the lesion in 91.6% (33/36) cases and adjacent to the lesion in 8.3% (3/36) cases |
| 8 | Nasu et al./201917 | Radial EBUS+GS | UM- S20-17S, UM-S20-20R, Olympus | 1.4, 1.7 | Conscious/deep sedation and topical anaesthesia | Yes, intubated 8.5mm ET | 1.9 | 3 | NA | 5...6 | 14.1mm2 (range=3.67...40.7) | 2.62mm2 (range=0.737...10.0) | Cryobiopsy with GS and positive bronchus sign were significantly associated with increased diagnostic yield of cryobiopsy (odds ratio (OR), 11.6; p=0.044 and OR, 21.5; p=0.034, respectively) |
| 9 | Udagawa et al./202021 | Radial EBUS ..GS+Fluoroscopy | NA | NA | Moderate sedation (Pethidine and Midazolam) and topical anaesthesia or GA | Yes, at anaesthesiologists discretion | 1.9 | 3...5 | 2 (1...5) | 5 (2...12) | 15mm2 (1...42) | 2mm2 (0.3...28) | Cryobiopsy yielded larger amounts of DNA (median: cryoprobe, 1.60..g vs forceps, 0.58..g, p=0.02) and RNA (median: cryoprobe, 0.62..g vs forceps, 0.17..g, p<0.01) extracted from samples, and tended to yield greater rates of PD-L1 expression >1% (51% vs 42%) |
ETT: endotracheal tube; GA: general anaesthesia; GS: guide sheath; R EBUS: Radial Endobronchial Ultrasound.
Quality assessment of the studies included in the systematic review and meta-analysis of cryobiopsy for PPL.
| MINORS tool for observational studies | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Serial no. | Question | Hibare et al. | Herath et al. | Taton et al. | Arimura et al. | Kho et al. | Imabayashi et al. | Nasu et al. | Udagawa H et al. |
| 1 | A clearly stated aim ..÷ the question addressed should be precise and relevant in the light of available literature | 2 | 1 | 2 | 2 | 2 | 2 | 2 | 2 |
| 2 | Inclusion of consecutive patients ..÷ all patients potentially fit for inclusion (satisfying the criteria for inclusion) have been included in the study during the study period (no exclusion or details about the reasons for exclusion) | 2 | 1 | 2 | 1 | 0 | 0 | 0 | 1 |
| 3 | Prospective collection of data ..÷ data were collected according to a protocol established before the beginning of the study | 0 | 2 | 2 | 2 | 0 | 0 | 0 | 2 |
| 4 | Endpoints appropriate to the aim of the study ..÷ unambiguous explanation of the criteria used to evaluate the main outcome which should be in accordance with the question addressed by the study. Also, the endpoints should be assessed on an intention-to-treat basis. | 2 | 1 | 2 | 2 | 2 | 2 | 2 | 2 |
| 5 | Unbiased assessment of the study endpoint- blind evaluation of objective endpoints and double-blind evaluation of subjective endpoints. Otherwise, the reasons for not blinding should be stated | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 |
| 6 | Follow-up period appropriate to the aim of the study- the follow-up should be sufficiently long to allow the assessment of the main endpoint and possible adverse events | 2 | 2 | 2 | 2 | 1 | 1 | 1 | 2 |
| 7 | A loss to follow up less than 5%- all patients should be included in the follow-up. Otherwise, the proportion lost to follow up should not exceed the proportion experiencing the major endpoint | 2 | 2 | 2 | 2 | 1 | 1 | 1 | 2 |
| 8 | Prospective calculation of the study size- information of the size of the detectable difference of interest with a calculation of 95% confidence interval, according to the expected incidence of the outcome event, and information about the level for statistical significance and estimates of power when comparing the outcomes | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 |
| 9 | An adequate control group- having a gold standard diagnostic test or therapeutic intervention recognized as the optimal intervention according to the available published data | 2 | 2 | 2 | 2 | 2 | 2 | 2 | 2 |
| 10 | Contemporary groups- control and studied group should be managed during the same time period (no historical comparison) | 2 | 2 | 2 | 2 | 2 | 2 | 2 | 2 |
| 11 | Baseline equivalence of groups- the groups should be similar regarding the criteria other than the studied endpoints. Absence of confounding factors that could bias the interpretation of the results | 2 | 2 | 2 | 2 | 2 | 2 | 2 | 2 |
| 12 | Adequate statistical analyses- whether the statistics were in accordance with the type of study with calculation of confidence intervals or relative risk | 1 | 0 | 2 | 2 | 2 | 2 | 2 | 2 |
| For every item | |||||||||
| ..úNot reported (0 points)..Ñ, ..úReported but inadequate (1 point), or ..úReported and adequate (2 points)..Ñ to judge | |||||||||
| MINORS (Score/Total) | 18 | 16 | 21 | 20 | 15 | 15 | 15 | 20 | |
| The MINORS tool consists of a checklist of eight items developed for non-comparative studies and four additional items for comparative studies. Items on the MINORS tool are scored as 0 (not reported), 1 (reported but inadequate) and 2 (reported and adequate), resulting in a total score of 16 for non-comparative studies and 24 for comparative studies. | |||||||||
| Cochrane tool for RCT | |
|---|---|
| Component | Schuhmann et al. |
| 1. Random sequence generation | Unclear risk |
| 2. Allocation concealment | Unclear risk |
| 3. Blinding of participants and personnel | High risk |
| 4. Blinding of outcome assessment | Low risk |
| 5. Incomplete outcome data | Low risk |
| 6. Selective reporting | Low risk |
| 7. Other sources of bias | Low risk |
For every item: ..úLow risk..Ñ, ..úHigh risk..Ñ, or ..úUnclear risk..Ñ to judge.
Artificial airways, if used, included endotracheal intubation with an endotracheal tube or rigid bronchoscopy. An artificial airway conduit (rigid bronchoscope or an endotracheal tube) was routinely used in five studies.4,9,17,18,20 Balloon blockers were variably used. The flexible bronchoscopes used for performing cryobiopsy had an internal channel diameter from 2.0mm to 3.2mm. Deep sedation, general anaesthesia or total intravenous anaesthesia (TIVA) were used to optimize patient comfort in different studies. In two studies, general anaesthesia was used routinely.4,20 Radial endobronchial ultrasound (R-EBUS) guidance with or without a guide sheath (size 1.4...1.7mm) was used in all studies for localization of PPL before the biopsy. Fluoroscopy was used in four studies.4,9,15,21 Guide sheaths, if used, for R-EBUS were1.95mm SG-200C or 2.55mm SG-201C (Olympus, Olympus Corporation, Japan). As the guide sheath was 2cm longer than the 1050mm long, 1.9mm flexible cryoprobe, Herath et al. trimmed the guide sheath by three cms from the distal end for to improve performance of biopsy.19 Electromagnetic navigation (EMN) was used in 1 study 20 and virtual bronchoscopic navigation (VBN) in 2 studies for pre-procedural planning.16,17 Most studies used a 1.9mm cryoprobe (ERBE, Germany) except one (used 1.2mm cryoprobe).4 Most of the studies used a freezing time between three to five seconds during cryobiopsy. Rapid onsite evaluation (ROSE) was used in two studies.16,18 Schuhmann et al. reported longer procedure time with cryobiopsy as compared to forceps biopsy (5.1..2.75min, forceps biopsy and 11.6..4.4min, cryobiopsy; p<0.0001). Kho et al. reported significantly increased diagnostic yield with cryobiopsy in eccentrically and adjacently orientated lesions 75.0%, compared to 48.8% with forceps biopsy (p<0.05). No difference was found in the yields in concentric lesions.9 Nasu et al. reported an increased diagnostic yield with cryobiopsy in the presence of the bronchus sign.17
Specimen qualitySpecimen area was reported in some studies, and where comparative data was available, specimen size was larger in cryobiopsy samples as compared to forceps biopsy. The specific metric used to denote size differed across studies from an area on slide or tissue volume. On artefact comparison on both cryobiopsy and forceps biopsy, no difference was observed in the semi-quantitative scoring for tissue morphology.4 In the context of lung cancer, a greater amount of DNA and RNA were extracted from the cryobiopsy samples, and there were higher success rates for RNA sequencing and whole-exome sequencing with the cryobiopsy samples.21 There was good concordance for TTF-1 and p40 immunostaining between the cryobiopsy and forceps biopsy samples. Cryoprobe biopsy samples also yielded greater rates of PD-L1 expression >1% on Immunohistochemical analysis.21
ComplicationsProcedure-related complications reported were mainly mild bleeding. Out of the 222 patients undergoing cryobiopsy (with reported complications data), there were three pneumothorax and one episode of severe bleeding. Sixty-five patients (65/222, 29.3%) experienced minor complications in the form of mild to moderate bleeding. Most of the bleeding events were mild that settled with either a prolonged bronchoscopic suction or local adrenaline instillation.4,9 One event of desaturation was also reported, which recovered when the procedure was stopped temporarily.15 Post procedure pneumonia was also reported (1 case).
Meta-analysisThe pooled diagnostic yields of of transbronchial cryobiopsy and forceps biopsy were 77% (95% CI, 71%...84%)(I^2=38.72%, p=0.11) (Fig. 2), and 72% (95% CI, 60%...83%)(I^2=78.56%, p<0.01) (Fig. 3), respectively. The comparative diagnostic yields were similar (RR1.05, 95% CI 0.96...1.15) (I^2=57.2%, p=0.017). (Fig. 4) The difference in diagnostic yield between cryobiopsy and forceps biopsy was 5% (95% CI, -6%...15%), and was not significant (Supplementary Fig. 1). There was the presence of significant heterogeneity [I2=57.2%, p=0.017]. There was no evidence of publication bias on the visual examination of the funnel plot, Eggers test (p=0.11) (Fig. 5).
The findings of this systematic review and meta-analysis indicate that R-EBUS guided transbronchial cryobiopsy is a safe and efficacious modality for bronchoscopic evaluation of PPL...s. The overall diagnostic yield of cryobiopsy for PPL was 77% and was not inferior to standard bronchoscopic forceps biopsy performed under R-EBUS guidance.
The development of Radial EBUS technology has revolutionized the bronchoscopic approach to sample peripheral pulmonary lesions.22 However, the yield of R-EBUS guided samples using conventional forceps is often suboptimal. This lower yield may be due to superficial sampling or crushing artefacts, and these samples may not be appropriate for immunohistochemistry or molecular analyses.9 The use of a cryoprobe for bronchoscopic biopsy allows one to obtain significantly larger samples. Schuhmann et al.4 reported an average tissue area of 11.17mm2 with cryobiopsy (4.69mm2 in the forceps biopsy group). In comparison, Udagawa et al.21 found an average tissue area of 15mm2 (2mm2 in the forceps biopsy group). Cryoprobe assisted biopsy from visible endobronchial growths provides a diagnostic yield superior to conventional forceps biopsy.23 Transbronchial lung cryobiopsy (TBLC) is commonly performed for the histological diagnosis of diffuse parenchymal lung diseases (DPLD).8 The diagnostic yield of TBLC for DPLD...s maybe comparable with video-assisted thoracoscopic surgery (VATS) lung biopsy with significant cost savings and reduced risk of mortality.24
The first study on the feasibility and utility of transbronchial Cryobiopsy for PPL was published by Schuhmann et al. in 2013.4 This study is the only published RCT comparing the diagnostic yield with forceps biopsy, and multicentre studies are lacking. Since then, few studies have highlighted the safety and feasibility of performing transbronchial cryobiopsy for PPL...s.
This meta-analysis has certain limitations. It included only one RCT and a relatively small number of prospective studies with uncontrolled variables. The strength of the meta-analysis is the fact that it is the only systematic review and meta-analysis on this subject. The major complication rate of cryobiopsy for PPL was low (1.8%), and this is less than that observed for TBLC for DPLD. The incidence of pneumothorax in TBLC for DPLD varies from 10% to 20% or even greater.24 The risk of clinically significant bleeding may also be greater.25 The severe bleeding rate in our meta-analysis (1/222, 0.4%) was low. Still, it is preferable that cryobiopsy for PPL...s be performed after securing the airway using either endotracheal intubation or rigid bronchoscopy to handle inadvertent complications, similar to the approach in DPLD...s.10 The use of a bronchial blocker to improve the safety of the procedure is also suggested.7 Procedural heterogeneity with varying freezing times, retrospective nature of many studies which may not have captured all complications limit the conclusions that can be drawn concerning complications. Still, the findings indicate that that cryobiopsy for PPL...s maybe a safer procedure than for DPLD.
In this analysis, transbronchial cryobiopsy for PPL was not inferior to forceps biopsy. This observation is contrary to observation regarding cryobiopsy for DPLD wherein the yield of cryobiopsy is superior to forceps biopsy.25 The possible reasons may be the operator expertise and more consistent localization of the lesion. Multicenter studies are the need of the hour to establish the utility of cryobiopsy for PPL in a real-life setting. The available studies did not employ a standardized protocol, and there were observed variations in the technique, anaesthesia methods and ancillary modalities. Future studies should also focus on standardization of the various technical aspects of the procedure like the use of artificial airways and occlusion balloons. The use of a guide sheath also needs further exploration as the use of a guide sheath was not routinely possible in most cases with the larger 1.9mm cryoprobe, as it could hinder this procedure. The 1.1mm cryoprobe appears an exciting addition and requires further exploration.26,27 This may allow the use of a radial EBUS in its full capacity with the maintenance of the over sheath. The more delicate probe can allow access to more distal locations and may allow sampling through a guide sheath contrary to the larger diameter cryoprobe. The use of a thinner cryoprobe may translate into possibly lower bleeding risk. These thinner cryoprobes may also be more manoeuvrable to perform upper lobe biopsies.28 However, it is unclear whether these biopsies with smaller cryoprobes will result in diagnostic yields similar to those described with larger probes.
High SUVmax values in PET-CT were associated with an increased diagnostic yield of transthoracic biopsy of lesions in the setting of lung cancer. Further studies should also examine these factors on the yield of cryobiopsy and conventional forceps bronchoscopic biopsy of PPL.29
ConclusionThe results of this meta-analysis demonstrate that transbronchial cryobiopsy is a safe and efficacious option for sampling peripheral pulmonary lesions. Although we did not observe a superior yield of cryobiopsy as compared with forceps biopsy for PPL, there is a need for multicentre studies to study the utility of this modality in real-life settings. The technique of transbronchial cryobiopsy for PPL also requires standardization.
Authors... contributionsSPB: Literature search, data abstraction, prepared manuscript draft, revised the manuscript.
SM: Data abstraction, revised the manuscript, data analysis.
PT, VH, AM, RG: Revised the manuscript.
KM: Conceived the idea, literature search, data abstraction, performed meta-analysis, prepared manuscript draft, revised the manuscript.
KM is the guarantor of this paper.
Funding sourcesNone.
Statement of ethicsThis study does not include human subjects for research as this is a meta-analysis of published studies.
Conflicts of interestThe authors have no conflicts of interest to declare.
NA.