Recurrence of TB has been associated with poor adherence to treatment, smoking, alcoholism, unemployment, drug abuse, the severity of pulmonary cavitation, HIV infection and duration of treatment.1 However, the risk factors can vary considerably across countries and between studies. Since little is known about risk factors for recurrence in Portugal, we aimed to identify predictors of treatment recurrence in the country. Surveillance data on TB for the period of 2002–2009 was provided by SVIG-TB, a database from the Portuguese National Health System. For this analysis, only cases of confirmed TB disease were considered, and patients that had information on the first and second TB episodes. The variables studied were chosen as TB risk factors according to previous reports about TB recurrence. The existence of missing data was assessed and missing data characterised (missingness between 0 and 10%). From this characterisation, we chose to explore multiple imputation using random-forest based on multivariate imputation by chained equations. We used a semi-parametric Cox regression model in which the event of interest was the second episode of TB, with time being measured from the end of treatment for a first TB episode. Patients without the second episode of TB before the end of the study were eliminated. A total of 8364 individuals were analysed, of which 145 (1.73%) had a recurrent TB episode during the time of the study.
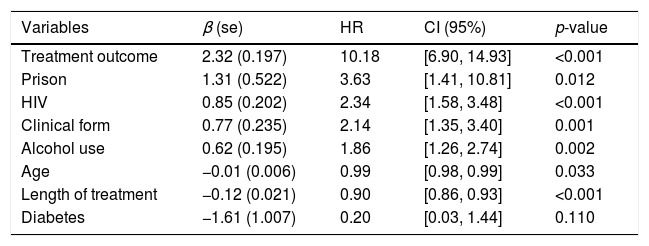
Patients who defaulted TB treatment are ten times more likely to suffer a recurrent case of TB (Table 1). As expected,1 HIV was positively associated with recurrence of TB disease (Table 1). Alcohol use disorders have been associated with recurrent TB, mostly by linkage to other confounding factors.1,2 We found that even when considering treatment default, TB patients with an alcohol use disorder still had 86% increase in the risk of TB recurrence (Table 1). Incarceration is a known risk factor for TB mainly due to overcrowding, delayed diagnosis and/or inadequate treatment. Our study shows that in Portugal, the risk of TB recurrence for prison inmates is four times higher (Table 1). The standard 6-month treatment regimen is often insufficient to prevent TB relapse,3 nevertheless, the study of treatment length as a risk factor has been mostly restricted to TB patients living with HIV.4 Longer treatments are usually prescribed for patients with poor prognosis, potentially confounding a beneficial effect. We found a decrease of 10% in the risk of recurrence per added month of treatment, even accounting for the effect of other risk factors (e.g. HIV), which may suggest a need to reevaluate standard treatment regimens (Table 1). The inclusion of the clinical form in a study about recurrence is unusual. Most studies discard extrapulmonary TB cases since this form is much less infectious, contributing less to overall TB epidemics.1 Nevertheless, we found that there is a two-fold increase in the risk of recurrence when suffering from an extrapulmonary form of the disease (Table 1). Driver et al.,4 showed that in TB patients living with HIV, extrapulmonary disease increased the risk of recurrence, and Millet et al.,2 suggested that this was true irrespective of HIV status. Interestingly, some TB risk factors have been shown to be associated with the clinical form – i.e. having HIV or being young increases the chance of having an extrapulmonary infection, while smoking and living with diabetes increases the chance of a pulmonary TB. Regardless of the association between youth and extrapulmonary TB, the role of age in TB recurrence is somewhat uncertain, with some studies5 indicating a decrease of risk for older people while others6 suggest a reduction for younger individuals. In Portugal, we found that an increase of one year in the age of the patient leads to a decrease of 1% in the risk of a recurrent episode (Table 1). The association between diabetes and TB incidence has been relatively established, but the association with TB recurrence is unclear.3 Although the variable is not significant, we have estimated a decrease of 80% in the risk of a recurrent episode when having diabetes (Table 1). In this study, 55% of recurrence occurred in the first 12 months after treatment completion, suggesting that in Portugal most of TB recurrence cases were due to relapse since relapse occurs not long after the end of treatment.7 Nevertheless, future studies should consider the inclusion of mycobacterial DNA information to distinguish between relapse and exogenous reinfection.
Hazard ratios and 95% confidence intervals for TB recurrence.
| Variables | β (se) | HR | CI (95%) | p-value |
|---|---|---|---|---|
| Treatment outcome | 2.32 (0.197) | 10.18 | [6.90, 14.93] | <0.001 |
| Prison | 1.31 (0.522) | 3.63 | [1.41, 10.81] | 0.012 |
| HIV | 0.85 (0.202) | 2.34 | [1.58, 3.48] | <0.001 |
| Clinical form | 0.77 (0.235) | 2.14 | [1.35, 3.40] | 0.001 |
| Alcohol use | 0.62 (0.195) | 1.86 | [1.26, 2.74] | 0.002 |
| Age | −0.01 (0.006) | 0.99 | [0.98, 0.99] | 0.033 |
| Length of treatment | −0.12 (0.021) | 0.90 | [0.86, 0.93] | <0.001 |
| Diabetes | −1.61 (1.007) | 0.20 | [0.03, 1.44] | 0.110 |
se, standard error; HR, hazard ratio; CI, confidence interval.
To the best of our knowledge, this is the only study, to date, covering risk factors for TB recurrence in Portugal. The study concerns the period 2002–2009 and, although the situation may have changed over the last decade, this cannot be assessed unless datasets linking multiple disease episodes at the individual level are made available to researchers. Understanding risk factors for TB recurrence in Portugal can help to define new guidelines to reduce the prevalence of recurrence, decreasing the chance of multi-drug resistant TB development.
FundingCristina Rocha's research is partially supported by FCT (Fundação para a Ciência e a Tecnologia), Portugal, through project UID/MAT/00006/2019. MGMG received funding from Fundação para a Ciência e a Tecnologia (IF/01346/2014).
Conflict of interestThe authors reported no conflict of interest.
We thank the members of the Collective Dynamics Group of Instituto Gulbenkian de Ciência for help and discussion at various points in the project. We also thank our collaborators from the Portuguese public health authorities, namely, Programa Nacional de Luta contra a Tuberculose, and the reference laboratories, Instituto Nacional de Saúde Ricardo Jorge and Administração Regional de Saúde de Lisboa e Vale do Tejo.