Systemic inflammation has been implicated in the pathogenesis of chronic obstructive pulmonary disease (COPD) systemic effects. However, most COPD patients do not suffer from persistent systemic inflammation even after exacerbations and exercise and scientific evidence has provided conflicting results. Our aim is to evaluate inflammatory gene expression at rest and at 1 and 24h after strenuous exercise in COPD patients and study the patient variables associated with inflammatory expression.
A cross-sectional study was conducted in COPD patients who were recruited on entry to a pulmonary rehabilitation (PR) program. Demographic, clinical and functional data were collected. Blood samples were collected and gene expression was analyzed by reverse transcriptase polymerase chain reaction for IFNg, IL1b, IL6, IL8, TNFa, TGFb1 and iNOS.
The study included 21 patients (15 men, 71.4%), mean age 66.1 years old (SD=8.27), mean FEV1 46.76% (SD 20.90%), 67% belonging to GOLD grade D, mean BODE index of 3.9, 90.5% with smoking history, mean BMI 25.81 (SD=4.87), median of 1.29 exacerbations in the previous year.
There was no statistical significant difference between inflammatory expression at rest and at 1h and 24h after the maximal exercise test for all tested genes.
We found an association between BMI and inflammatory expression at all the points of time checked, a slight inverse association occurs with low BMI for mRNA IL1b, IL6, TNFa, TGFb1 and iNOS, and there was a more pronounced positive association for obese patients for all tested genes.
This preliminary study did not show an enhanced inflammatory gene expression from rest to 1h and 24h after short-term exercise, but did show an increased inflammatory gene expression in both BMI extremes, both at rest and after exercise, suggesting not only malnourishment, but also obesity as potential links between COPD and systemic inflammation. Studies with larger samples and designed to definitely exclude OSA or OHS as confounding factors in obese patients are required.
Chronic obstructive pulmonary disease (COPD) is associated with important extrapulmonary manifestations, including weight loss, skeletal muscle dysfunction, cardiovascular disease, depression, osteoporosis, reduced exercise tolerance, and poor health status.1–3 Although the pathobiology of COPD has not been fully determined, persistent systemic inflammation has been implicated in the pathogenesis of the majority of these systemic effects.3,4 Elevated circulating levels of white blood cells, C-reactive protein (CRP), interleukins 6 (IL-6) and 8 (IL-8), fibrinogen and tumor necrosis factor alpha (TNFa) have been reported in patients with COPD.5 However, the ECLIPSE study5 has demonstrated that a large group of patients with COPD do not suffer from systemic inflammation.
The role of exercise in COPD inflammatory process has also been a matter of debate.6–15 Patients with COPD are exposed to a systemic inflammation that is amplified by exhaustive exercise. Inflammatory response to exercise is more pronounced in patients with COPD when compared to healthy controls, even at lower levels of exercise intensity.9,15 However, scientific literature on this subject is also not consensual, as several studies have shown a reduction in the level of TNFa protein expression in COPD subjects.10 As pointed out by Canavan and colleagues,11 some of the heterogeneity of these results might be caused by the different methods that were used in the studies (patient characterization, exercise protocols, and assay techniques). Crul and colleagues12 did not find any evidence of muscle inflammation in patients with COPD, independently of whether they were in a stable or an acute exacerbation state. Conversely, others have suggested an anti-inflammatory effect of regular exercise in some low systemic inflammation chronic diseases, with beneficial outcomes in disease prevention and symptomatic improvement.6,13,14
In this study we aim to evaluate the inflammatory and immune regulatory gene expression profiling in peripheral blood determined at rest in patients with COPD, and the possible modification after strenuous exercise, and search for variables and patients’ characteristics associated with inflammatory expression.
Material and methodsPatientsA cross-sectional study was conducted on a sample of 21 patients diagnosed with COPD, according to the Global Initiative for Obstructive Lung Disease Project (GOLD)16 as post bronchodilator FEV1/FVC<0.70. Patients were consecutively recruited on entry to a pulmonary rehabilitation (PR) program at our PR Unit, from January to December 2010. Participants were selected if clinically stable in the previous four weeks and able to exercise and to answer health status questionnaires. Patients diagnosed with other significant lung diseases, e.g. asthma, bronchiectasis or other conditions that might cause dyspnea or affect exercise performance, were excluded.
Data collectionData collection included age, body mass index (BMI), smoking history, number of exacerbations in the previous year and comorbidities. Clinical data were obtained by interview and from medical records, including the review of concomitant medications. Charlson,17 Charlson-age18 and COTE (COPD specific comorbidity test)19 indexes were calculated based on comorbidities data. Participants completed questionnaires on dyspnea (modified Research Council breathlessness scale and Mahler’ baseline dyspnea index),20 activities of daily living (London Chest Activity of Daily Living scale – LCADL),21 anxiety and depression (Hospital Anxiety and Depression scale – HADS),22 and health status (St. George's Respiratory Questionnaire – SGRQ).23
Pulmonary function data were obtained using standardized equipment (SensorMedics Corporation, Yorba Linda, CA, USA). Post-bronchodilator spirometric values were obtained. Data were measured as absolute values (L) and as percent predicted of reference values.
Exercise test and laboratory proceduresPatients were subjected to an incremental exercise test to maximum tolerated on a treadmill or on a cycle ergometer (Fig. 1). Treadmill protocol started with a three minutes warming up at 2.0km/h and 0° inclination, followed by 0.5km/h increments per minute and 0° inclination until the patient attained a brisk walking speed without running, and then increments of 2° inclination every minute until exhaustion. Cycle ergometer protocol starts with a three minutes warm up with no added resistance, followed by 10W of increments each minute until exhaustion. Safety criteria for terminating the exercise test were applied according to ATS/ACCP guidelines.24
Whole blood samples were collected from each patient at three different time points: at rest (T0), and at one hour (T1) and 24h (T2) after the exercise test. Additionally, a blood sample from the first ten patients entering the study was collected the day before (C0), and considered as a calibration sample for relative gene expression analysis to establish the expression pattern of target genes in resting condition. These participants were told to maintain normal low-intensity activities of daily living and avoid exercising above that intensity in the previous 48h.
RNA integrity from blood cells was immediately preserved at collection with the PAXgene Blood RNA Tubes (PreAnalytiX GmbH, Hombrechtikon, Switzerland). Each sample tube was kept at room temperature for 2h, followed by incubation at −20°C for 24h and thereafter stored at −80°C. Thawed tubes were processed with the PAXgene Blood RNA Kit (PreAnalytiX GmbH) for isolation of total RNA according to manufacturers’ instructions, including DNAse digestion. Yield of purified RNA samples was evaluated with a NanoDrop 2000 spectrophotometer (Thermo Fisher Scientific, Wilmington, USA) and stored at −80°C. Gene expression was analyzed by reverse transcriptase polymerase chain reaction (RT-PCR), essentially as previously described.25 Briefly, a template cDNA was generated by reverse transcription from 1 to 2μg of total RNA using the High Capacity cDNA Archive Kit (Applied Biosystems, Foster City, CA). Measurements of target genes and endogenous control (beta actin) were performed using the Taqman Gene Expressions Assays in combination with TaqMan Fast Advanced Mastermix on a 7900 HT system (Applied Biosystems), according to manufacturers’ instructions. The analyzed genes and each primer/probe assay ID were the following: interleukin 1 beta (IL-1β, Hs00174097_m1), interleukin 6 (IL-6, Hs00174131_m1), interleukin 8 (IL-8, Hs00174103_m1), inducible nitric oxide synthase (iNOS, Hs00167248_m1), interferon gamma (IFN-γ, Hs00174143_m1), tumor necrosis factor (TNF-α, Hs00174128_m1), transforming growth factor beta 1 (TGF-β1, Hs00998133_m1). The efficiency for each primer/probe assay was above 95% (as determined by the manufacturer).
Endogenous gene expression was used for each assay normalization and gene expression was calculated by the adapted formula 2−DCt*1000, which infers the number of mRNA molecules of the gene of interest per 1000 molecules of the endogenous controls.26 DCt stands for the difference between the cycle threshold of the target gene and that of the endogenous control genes.
A week after the maximum exercise test, patients performed a 6-minute walking test (6MWT), standardized according to international guidelines.27 6MW distance, FEV1% predicted after bronchodilator, mMRC dyspnea scale and BMI data were aggregated to calculate BODE index.28
Subjects were willing and able to participate in this study and gave written informed consent prior to baseline measurements. The hospital's Ethics committee and administration board approved the trial (IRB: Study 25/07_CE/027/07), and all data were processed anonymously according to the institution's privacy policy.
Statistical analysisCategorical data were presented as frequencies and percentages, and continuous variables as mean or median, SD or interquartile range: 25th percentile (P25) to 75th percentile (P75). To verify the normality assumption of parametric tests, Shapiro–Wilk goodness-of-fit test and Q–Q plots were used. To compare genetic inflammatory markers between T0 and C0, T0 and T1, and T0 and T2, nonparametric tests were used (exact Wilcoxon signed ranks test or sign test when the differences had no symmetric distributions). To identify associations between patients’ characteristics and genetic inflammatory expression in all time points, LOWESS (Locally Weighted Scatterplot Smoother), Spearman's correlation coefficient and Mann–Whitney test were applied. The significance level α=0.05 was considered. Due to the exploratory nature of the study, no multiple testing procedures were used.
All data were analyzed using the Statistical Package for the Social Sciences for Windows 21.0 (IBM Corp. Released 2012. IBM SPSS Statistics for Windows, Version 21.0. Armonk, NY: IBM Corp.).
ResultsA total of 21 patients were recruited, agreed to participate and entered the study. There were no dropouts. Participants sample included 15 men (71.4%), with a mean age of 66.1 years (SD=8.27), 19 (90.5%) with smoking history (16 previous smokers – 76.2% and 3 active smokers – 14.3%), with an average 69 pack-years (range 12–150) and 2 never smokers (9.5%). These latter patients were in fact never smokers with a long-term second-hand smoke exposure. One woman was exposed for years to her husband smoking at home, and the other woman was also exposed for years to her family smoking at home and to her colleagues smoking at work. Fourteen patients (66.6%) had respiratory failure, 54.1% hypoxemic and 9.5% hypercapnic.
According to GOLD categories,16 14 (67%) patients belonged to category D. Mean FEV1 was 46.76% (SD=20.90%,min 21%, max 97% predicted), median BODE was 3.9 (min 0, max 8) and a median of 1.3 exacerbations (min 0, max 10) had occurred in the previous year.
Mean BMI were 25.81kgm−2 (SD=4.9), ranging from 17 to 34kgm−2. Three participants (14%) were malnourished, 8 (38%) were overweight, and four patients (19%) were obese (BMI 30–34kgm−2). Of these latter, one had mild obstructive sleep apnea (OSA) with an Apnea Hypopnoea Index (AHI) of 7.1 events/hour, and the other had moderate obstructive sleep apnea with an Apnea Hypopnea Index (AHI) of 25.7 events/hour. Neither of these two patients were on CPAP at the time of the study. In the other two obese patients, OSA or hypoventilation were excluded based on the absence of typical night and daytime complaints, on physical examination and on normal arterial blood gases.
In relation to respiratory comorbidities, tuberculous sequelae diagnosis (n=7) were made according to a history of past pulmonary tuberculosis and imagiologic features such as calcified granuloma, focal fibrosis or localized pleural thickening. None of these radiologic features were currently considered as clinically relevant. Similarly, cylindrical bronchiectasis was found in two patients, and interpreted as associated with COPD.
The most prevalent comorbidities in our sample were cardiovascular (CV) disease, arterial hypertension being the most common (52.4%).
Both patients with and without cardiovascular comorbidities exercised on the same ergometers (cycle and treadmill) and used the same protocols: of 15 patients with cardiovascular comorbidity, 5 exercised on cycle ergometer and 10 on treadmill. Of 6 patients without cardiovascular comorbidity, 3 exercised on cycle ergometer and 3 on treadmill.
Patients without cardiovascular comorbidity achieved higher work load on cycle ergometer [mean (SD): 83 (22)W, min–max: 70–108W versus mean (SD): 58 (25)W, min–max: 40–100W] but lower workload on the treadmill [mean (SD): 5 (0.76)km/h and 7 (6.93)° inclination, min–max: 4.5km/h, 2.2° – 6km/h, 12° versus mean (SD) 5 (0.96)km/h and 10° (7.44), min–max: 4.0km/h, 0° – 6km/h, 22°]. All the patients exercised to their maximal tolerance.
Cardiopulmonary exercise tests data are shown in Annex, Table 1a.
Mean 6MWD was 346.1 meters (SD=86.1), ranging from 185 to 520 meters. Oximetry lowest values at 6-minute walking test were on average 85%, ranging from 68% to 94%. Baseline characteristics are presented in Table 1.
Baseline characteristics of COPD participants. Spirometry values displayed are post-bronchodilator.
| Demographics | |
| Gender m/wn (%) | 15 (71.4%)/6 (28.6%) |
| Ageyears mean (SD) min–max | 66.05 (8.27) 50–82 |
| BMIkg.m−2 mean (SD) min−max | 25.81 (4.87) 17–34 |
| Pack'smean (SD) min–max | 69.05 (30.85) 12–150 |
| Symptoms/health status | |
| mMRCmedian (min–max) | 1.00 (0–3) |
| Mahler's BDImedian (min–max) | 7.00 (3–10) |
| LCADLmedian (min–max) | 19.15 (8–36) |
| HADS-anxietymedian (min–max) | 6.19 (1–14) |
| HADS-depressionmedian (min–max) | 5.62 (0–12) |
| SGRQmedian (min–max) | 45.95 (27–77) |
| BODEmedian (min–max) | 3.90 (0–8) |
| Exacerbations previous yearmedian (min–max) | 1.29 (0–10) |
| GOLD categories A/B/C/Dn (%) | 2 (9.5)/2 (9.5)/3 (14.3)/14 (66.6) |
| Comorbidities | |
| COTEmedian (min–max) | 1.24 (0–8) |
| Charlsonmedian (min–max) | 1.62 (1–3) |
| Charlson-agemedian (min–max) | 3.57 (2–5) |
| Respiratory failure/hypoxemic/hypercapnicn (%) | 14 (66.6)/12 (54.1)/2 (9.5) |
| Cardiovascular comorbiditiesn (%) | 15 (71.4) |
| Arterial hypertensionn (%) | 11 (52.4) |
| Congestive heart failure/Cor pulmonalen (%) | 5 (23.8) |
| Ischemic heart diseasen (%) | 2 (9.5) |
| Arrythmiasn (%) | 2 (9.5) |
| Tuberculous sequelaen (%) | 7 (33.3) |
| Bronchiectasisn (%) | 2 (9.5) |
| Obesity or overweightn (%) | 12 (57.1) |
| Malnourishedn (%) | 3 (14.3) |
| Diabetes mellitusn (%) | 1 (4.8) |
| Dyslipidemian (%) | 4 (19.0) |
| Osteoarticular pathologyn (%) | 4 (19.0) |
| Physiology | |
| FVCL mean (SD) min–max | 2.75 (0.95) 0.84–5.31 |
| FVC%mean (SD) min–max | 85.52 (23.74) 32–139 |
| FEV1L median (P25-P75) min–max | 1.02 (0.73–1.46) 0.47–3.25 |
| FEV1% mean (SD) min–max | 46.76 (20.90) 21–97 |
| FEV1/FVC% mean (SD) min–max | 43.48 (14.11) 21–69 |
| TLCL (n=19) mean (SD) min–max | 7.11 (1.37) 4.14–8.71 |
| TLC% (n=19) mean (SD) min–max | 121.53 (15.64) 86–155 |
| RVL (n=19) mean (SD) min–max | 4.26 (1.27) 2.51–6.43 |
| RV% (n=19) mean (SD) min–max | 189.79 (49.19) 128–293 |
| DLCO% (n=17) mean (SD) min–max | 45.12 (19.07) 16–95 |
| KCO% (n=17) mean (SD) min–max | 41.41 (17.49) 13–76 |
| PaO2 mmHg mean (SD) min–max | 65.46 (8.58) 50–77 |
| PaCO2 mmHg median (P25-P75) min–max | 42.10 (37.20–46.05) 35–60 |
| 6MWDm mean (SD) min–max | 346.05 (86.06) 185–520 |
| 6MWD%pred mean (SD) min–max | 61.00 (19.50) 29–102 |
| 6MWT SpO2% mean (SD) min–max | 84.57 (6.67) 68–94 |
Pharmacological treatment was as follows: 18 participants were on triple therapy with long-acting muscarinic antagonist (LAMA) Tiotropium, long-acting beta-adrenergic (LABA) Salmeterol or Formoterol and inhaled corticosteroid (ICS) Budesonide, Beclometasone or Fluticasone; 2 participants were on LABA and ICS, one was on LABA, ICS and short acting muscarinic antagonist (SAMA) Ipratropium and one participant was on rescue short acting beta-adrenergic (SABA) Salbutamol, and Acetylcystein. Eight patients were on Theophylline, 4 were on statins (Simvastatin, Atorvastatin or Rosuvastatin) and 2 were on Amiodarone.
Seven patients were on long-term oxygen therapy (LTOT) and one was on LTOT and non-invasive ventilation (NIV). One normal-weight patient was on CPAP due to obstructive sleep apnea.
The pharmacological therapy used by the four obese patients and the other participants was essentially the same: three were on triple therapy LAMA, LABA and ICS, one was on LABA and ICS. Three were on theophylline and 2 were on statins. None of the patients were on oral corticosteroids or Roflumilast. None of the four patients were on LTOT, NIV or CPAP.
Laboratory resultsIn the first 10 patients studied, we did not find a statistically significant difference between C0 and T0, the two time points of baseline resting condition (Annex-Table 2a). The characteristics of these first ten patients entering the study were similar to all group characteristics: 6 men (60%), 4 women (40%), mean age of 63.4 (SD: 6.9) years old (ranging from 56 to 79 years), mean post-bronchodilator FEV1%predicted 44.5% (SD: 23.6) (R: 21–85%), mean BMI 24.8kgm−2 (SD: 4.3) (R: 19–31kgm−2), mean 69.9 pack-years (SD: 20.9) (R: 45–100 pack-years).
In the entire sample, there was no statistically significant difference between inflammatory expression at rest and at 1h and 24h after the maximal exercise test (Table 2).
Median (P25–P75) of the mRNA inflammatory genes at rest (T0), and at 1h (T1) and 24h (T2) after maximal exercise test. Comparison of T1 versus T0 and T2 versus T0.
| T0 | T1 | T1 vs T0 p | T2 | T2 vs T0 p | |
|---|---|---|---|---|---|
| IFNg | 0.16 (0.04–1.57) | 0.19 (0.06–0.95) | 1.000b | 0.15 (0.05–1.17) | 0.739a |
| IL1b | 15.46 (6.42–266.69) | 19.38 (7.39–268.48) | 0.383b | 23.15 (5.63–250.58) | 0.664b |
| IL6 | 0.33 (0.11–2.81) | 0.51 (0.23–2.67) | 0.189b | 0.59 (0.10–2.05) | 0.815b |
| IL8 | 1.82 (0.97–69.57) | 4.70 (1.87–43.26) | 0.189b | 2.07 (0.94–43.37) | 0.664b |
| TNFa | 2.99 (2.55–47.00) | 3.36 (2.25–29.61) | 1.000b | 3.42 (2.51–38.21) | 0.383b |
| TGFb | 199.71 (64.86–5462.36) | 224.38 (63.91–4456.02) | 0.383b | 373.45 (75.18–5250.38) | 0.078b |
| iNOS | 0.03 (0.01–0.29) | 0.04 (0.01–0.60) | 0.791b | 0.04 (0.01–0.31) | 0.398a |
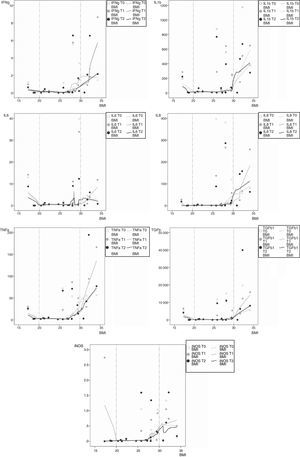
Looking at associations between patients’ characteristics and inflammation, the only evidence of an association was found between BMI and inflammatory expression at all time points. As presented in Fig. 2, a slightly inverse association occurs with low BMI (values under 20kgm−2; n=3) for inflammatory genes mRNA IL1b, IL6, TNFa, TGFb and iNOS. Moreover, a more pronounced positive association was found for obese patients (BMI above 30kgm−2; n=4) for all inflammatory mRNA genes tested, at all time points. No correlation coefficient estimates with corresponding p values were reported because only three patients were malnourished and four patients were obese.
We found no associations between inflammatory expression and all other patients’ characteristics and variables, including demographics, symptoms/health status, comorbidities and physiologic parameters (data not shown).
Due to the previously reported potential influence of cardiovascular comorbidity in the inflammatory expression of COPD patients,29 we looked for the association between this variable and inflammatory gene expression. Higher T1-T0 and/or T2-T0 differences were found (sometimes with statistical significance) for patients without cardiovascular comorbidity (Annex-Table 3a).
DiscussionIn our COPD patient sample, real-time polymerase chain reaction of the target mRNA inflammatory genes did not show increased inflammation either at rest or after a maximum exercise test. When looking at the inflammatory expression measured in resting samples on two consecutive days (control group), there were also no statistical differences between the two time points, which might reflect stability at rest mRNA parameters.
Persistent systemic inflammation is not a universal finding in patients with COPD. ECLIPSE study recently showed that in 1755 COPD patients, about 30% do not have systemic inflammation, and only a minority (16%) have persistent inflammation during 1 year follow up.5
Although we expected a significantly different mRNA inflammatory expression at 1 and at 24h after maximal exercise, in line with other authors, such as Van Helvoort et al.,15 who demonstrated that COPD patients, when compared to healthy subjects, are exposed to systemic inflammation that is intensified by exhaustive exercise, we did not find a significantly different mRNA inflammatory expression after short-term exercise.
However, in the subgroups of malnourished and obese COPD patients, we found opposite associations with mRNA inflammatory expression. When considering malnourished patients, mRNA levels of iNOS, IL6, IL1b, TNFa, and TGFb1 showed a tendency to decrease with increasing BMI values toward normal. Conversely, overweight and obese patients showed higher mRNA levels of TNFa, IFNg, IL1b, IL8, TGFb1, iNOS and IL6 as their BMI increasing above normal values. When considering all sample data, higher mRNA values of the malnourished on one side, and overweight and obese patients on the other, shown graphically in Fig. 2, might be blurred by the lower mRNA values of normal weight patients. This could explain the absence of a significant association of inflammation and exercise when taking into account the median values of all sample data.
ECLIPSE study also evidenced high BMI as one of the independent risk factors for persistent inflammation, both at baseline and at one year of follow-up.5 This association was not evident for fat free mass index, which suggests an important role for adipose tissue in systemic inflammation. Garcia-Aymerich and colleagues30 also identified a “systemic” COPD subtype characterized by a higher proportion of obesity in 342 COPD patients with a significant systemic inflammation; this pattern was maintained through a 4 years’ follow-up period. In the study of Tamakoshi and colleagues, an acute phase response evidenced by serum levels of C-reactive protein increased in obesity and was associated with insulin resistance.31
Most of the scientific evidence has previously shown that peripheral muscle atrophy and cachexia are associated with systemic inflammation in patients with COPD, when compared to patients with no muscle wasting.32,33 In the same way, in our study, malnutrition was also associated with higher expression on mRNA genes iNOS, IL6, IL1b and TGFb1, but to a lesser extent, and in the opposite direction, when compared to what happened in patients with higher BMI.
Inflammation in COPD has been associated with a worse prognosis.34 In the ECLIPSE study, persistent inflamed patients had significantly increased all-cause mortality and exacerbation frequency compared with non-inflamed patients.5 In our study, 8 patients were deceased after 4 years (38%), of whom, 4 (50%) were obese or overweight, and 3 (37.5%) were malnourished. Only one patient who died had had a normal BMI at baseline (data not shown).
Cardiovascular disease is the most prevalent comorbidity in COPD,35–37 and it has previously been associated with systemic inflammation (elevated C-reactive protein) in patients with COPD.29 However, although prevalent (71%), cardiovascular disease was not associated with higher inflammatory expression for all genes studied. These results do not seem to be influenced by different exercise protocols involved, as both patients with and without cardiovascular comorbidities exercised on the same ergometers, used the same protocols and exercised to their maximal tolerance. Patients without cardiovascular comorbidity achieved higher work load in cycle ergometer, but lower workload on the treadmill. However, assuming that the sample studied is too small to be representative, a definite conclusion of cardiovascular comorbidity not being associated with higher levels of inflammation after exercise in COPD patients can only be established with larger samples.
There is still an unmet need to characterize different COPD phenotypes that might benefit from different treatment approaches and respective outcomes. Inhaled corticosteroids, as an example, have limited efficacy in the reduction of systemic inflammation in COPD.38 On the other hand, a non-pharmacological approach, such as pulmonary rehabilitation with exercise training, has been proven to improve exercise tolerance, while modifying body composition toward a higher fat-free mass, improving muscle function, and, as evidenced by some studies, it might also attenuate systemic inflammation.6
The present study has a number of limitations that need to be addressed. A small sample size was not able to rule out the presence of an increased systemic inflammation. Although small, this sample size was enough to show graphically an association between BMI and inflammation. It is an exploratory study and longitudinal studies with larger samples are needed to confirm this association. Studies designed to definitely exclude OSA or OHS as confounding in obese patients are also required.
Another limitation might be the fact that levels of plasma proteins and related mRNA precursors are sometimes not coincident.39,40 Cytokine protein production is strictly regulated at multiple steps, including transcription (mRNA chain generation) and translation (protein synthesis from RNA). Plasma cytokine expression will depend on the efficiency of translation of mRNA into useful proteins.
Although our study suggested an association between obesity and several inflammatory genes, and other authors also evidenced RT-PCR as an attractive method of studying the gene expression of cytokines in whole blood,41 an additional study should be carried out to confront mRNA genes and the corresponding plasma proteins.
ConclusionsThis preliminary study evidenced an association between BMI (malnutrition and obesity) and the levels of inflammatory mRNA cytokine precursors, suggesting not only malnourishment, but also obesity as potential links between COPD and systemic inflammation.
Ethical disclosuresProtection of human and animal subjectsThe authors declare that no experiments were performed on humans or animals for this study.
Confidentiality of dataThe authors declare that they have followed the protocols of their work center on the publication of patient data.
Right to privacy and informed consentThe authors have obtained the written informed consent of the patients or subjects mentioned in the article. The corresponding author is in possession of this document.
Conflict of interest statementNone of the authors has a financial relationship with a commercial entity that has an interest in the subject of this manuscript.
Acquisition of laboratory analysis kits and lab ware were partially funded by a scholarship from Nova Medical School for research projects undertaken in the affiliated hospitals of NOVA Medical School.
We would like to thank Eunice Silva for her help in collecting blood samples and performing laboratory analysis.
Study was carried out at the Pulmonary Rehabilitation Unit, Pneumology Service, CHLN-Hospital Pulido Valente and in South Luso-Transplant Centre.